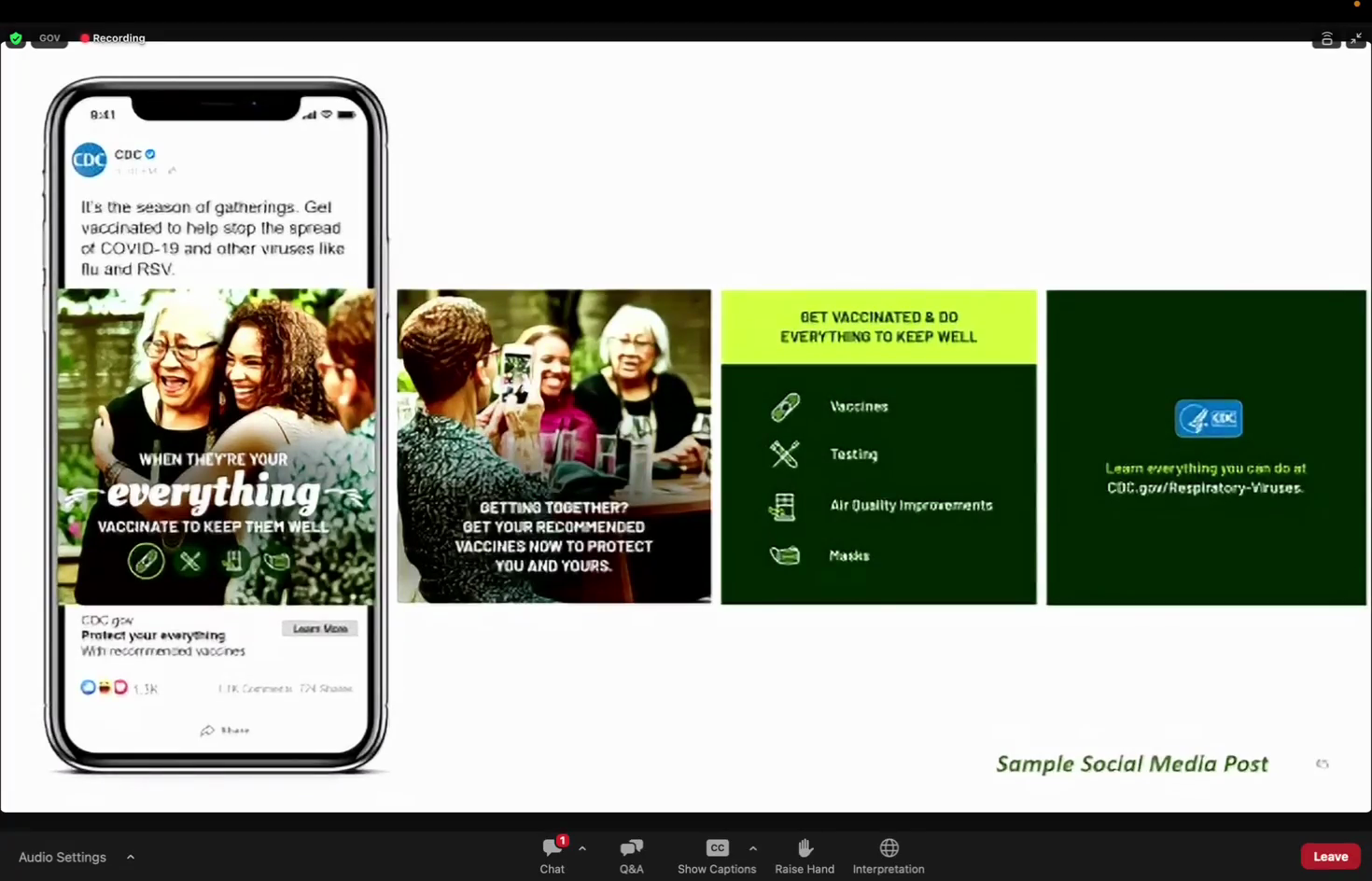
Alexander Tin's notes
tinalexander.github.io / notes / 2023 / 10 /
Learn more about this site and how to get in touch.
October 31, 2023
FDA meeting on Vertex’s exa-cel for sickle cell disease
- Source: webcast by Food and Drug Administration
- Date: broadcasted October 31, 2023
Nicole Verdun, FDA [09:18:45]
As many of you know, sickle cell disease is a debilitating hemoglobinopathy with significant unmet medical need and can carry a reduction in overall survival for those affected.
In addition, curative options are significantly limited.
I’ve had the pleasure of taking care of several sickle cell patients and admire the courageous and resilient patient community.
I’m also reminded of the sickle cell disease patient focused drug development program at FDA in which we heard directly from patients and their caregivers, which highlighted the significant unmet need in this disease.
It is an exciting time in cell and gene therapy that we are beginning to address some of this unmet need in a variety of diseases.
Exa-cel has been studied for treatment of sickle cell disease with severe vaso-occlusive crises and has shown efficacy and safety in this population.
In today’s discussion, we would like to focus the discussion specifically on the off target analyses for genome editing for exa-cel.
…
Daniel Bauer, Boston Children’s Hospital [10:05:14]
I would emphasize to patients that there’s often an uncertainty about the functional significance of off target edits that, you know, only a small part of the human genome actually codes for genes. Most of the human genome is noncoding. Its functional importance could be regulatory, but it’s likely that many places in the human genome can tolerate an off target edit and not have a functional consequence.
The challenge is we just don’t know for sure. And the only way to know that is careful follow up, I would say.
What I would emphasize to patients is what’s obvious to them, the known risks of the disease, that this is a terrible disease and that has to play in and then the risks of the therapy, which are known, which include things like the busulfan conditioning that’s used or whatnot.
So I would say this: my guess is it’s a relatively small risk in the scheme of this risk benefit, but it’s new, it’s unknown, but it’s easily measurable. And that’s one of the goals I would say of doing this under very careful circumstances is to try to learn what that risk is, so that we can continually improve those therapies.
…
Lisa Lee, CTGTAC [10:06:47]
I mean, theoretically, is it catastrophic?
Daniel Bauer, Boston Children’s Hospital [10:06:51]
In theory, as Dr Urnov said, there– these cell products have lots of cells in them, hundreds of millions of cells, and any one cell that goes awry could cause leukemia.
Now, has that ever been shown, that an off target effect of gene editing cause leukemia? No.
So theoretically, could it? Yes. Is there any evidence to suggest that it does? I would say no.
But we– this hasn’t really been done very much. We need to be humble and open to learning from these brave patients who are participating.
…
Taby Ahsan, CTGTAC [10:25:48]
I think for the rest of the day, it sets the stage to think about two things, which is: when is enough theoretical data sufficient to support a patient specific risk assessment?
And also, to your point, Dr. Urnov, which is: where are we in that curve of risk mitigation? And have we actually started getting to that asymptote, or is there more work to be done?
Those are two very important questions. And while we will start the discussion today, I think that this will continue to evolve over time.
…
Stephanie Krogmeier, Vertex [10:39:46]
The results from the exa-cel program in severe sickle cell disease are unprecedented.
Exa-cel demonstrated transformative efficacy, a strong safety profile, and a highly positive benefit risk for patients with severe sickle cell disease.
…
David Altshuler, Vertex [10:59:30]
The DNA sequence must match both the guide RNA and a short adjacent sequence known as the protospacer adjacent motif or PAM.
If the on target site is unique in the genome, as depicted on the left with a yellow dot, and if the guide is highly specific, then editing will occur only at the on target site.
However, if one were to choose an on target DNA sequence that is present at multiple genomic locations as depicted on the right in red, and one designed a guide that binds promiscuously at many places in the genome, then off target editing can occur.
Based on this understanding, three strategies to minimize the risk of off target editing are first to limit exposure to CRISPR.
Second, to select an on target site that is unique in the genome.
And third to optimize the guide RNA, not only for efficacy, but also for specificity.
From the start of the exa-cel program eight years ago, we were focused on minimizing and assessing the risk of off target editing.
…
David Altshuler, Vertex [11:00:35]
The design of exa-cel was shaped by three strategies to minimize off target risk.
First, we use an ex-vivo approach and transiently express CRISPR only in cells of the hematopoietic lineage.
Second, we selected the on target site in an intron of BCL11A that has a unique sequence with no other match in the human genome.
Third, we screened hundreds of candidate guide RNAs to select an optimal guide RNA that is no other match elsewhere in the human genome.
Now having designed exa-cel to minimize potential for off target editing, we then systematically evaluated the risk of off target editing using multiple orthogonal methods to detect potential off target edits, including sites nominated based on human genetic diversity and performing risk assessments as appropriate.
And the conclusion is that the design of exa-cel minimized potential for off target risk and multiple systematic evaluations did not identify evidence of off target editing by exa-cel.
…
Bill Hobbs, Vertex [11:45:56]
Our approach to this, if you boil it down is really that to do close clinical monitoring and follow up, which we recommend and do for all patients in the clinical study, irrespective of the variant or not.
And the rationale for the approach is that we know we have established a strongly positive benefit risk for this in a patient population with severe unmet need, who not only have a disease that is impacts quality of life, but also shortens their life as the disease relentlessly progresses.
The approach for that Dr. Altshuler described for the non-clinical package, which didn’t identify any specific off target risks, and did a risk assessment of any additional variant that could potentially occur in a patient, concluded that there was a low risk of a functional consequence to a patient. And therefore we had neither an off target to follow nor a specific variant of concern for a clinical outcome.
And we concluded from that, that the appropriate approach for all patients in the clinical study was close and careful clinical monitoring, which is independent of whether they have the variant or not, and assumes that any particular patient could have an off target effect, which we could then pick up in that process.
We also collected laboratory samples from both before and after treatment that would allow us to go and then subsequently investigate, should there be the need arise.
And so this is also reflected in our pharmacovigilance plan that Dr Simard described.
And so our approach has been to do careful close clinical monitoring, which was also referred to in the session this morning as the appropriate approach for all patients who receive a genetic therapy like exa-cel.
Gil Wolfe, CTGTAC [11:47:35]
So there’s no plan for molecular follow up, to look at editing at this off target site? It seems like there is quite a bit that could be learned with regards to off target editing rates in your treated patient population by looking retrospectively at editing at this site, now that you’ve treated more than 45 patients.
Stephanie Krogmeier, Vertex [11:47:58]
I will ask Dr. Altshuler to address your question.
David Altshuler, Vertex [11:48:06]
It’s an important question, and we’ve thought deeply about it. And the way we think about it is first taking into consideration, not only the off target assessment we’ve done, but all the other assessments and the package of data, and as Dr. Hobbs described, we don’t believe from the totality of data that we’ve collected, that additional nonclinical studies are gonna be informative.
And then we believe for clinical studies, the important thing is to follow all the patients, see if any events occur and then we’ll have the samples and the data to try and understand those events. And that’s our approach.
…
David Altshuler, Vertex [11:54:19]
I’d like to just go back in answering your question to the transient nature of CRISPR editing with exa-cel.
So the cells obviously are harvested, and then in the manufacturing process, they are briefly exposed to the CRISPR-Cas9 enzyme using a ribonucleoprotein particle, RNA protein complex, and then that is a short duration of editing, and then it’s gone.
So I guess if your question again, as Dr. Hobbs said, we do not imagine the need for nor intend there to be another treatment.
But just as your hypothetical, if it were– the exposure to CRISPR-Cas9 is extremely brief and not in the body of the person who has the cells, because it all takes place in the manufacturing process, and is then gone and does none of it residually is there and makes it into the patient.
…
Christopher Breuer, CTGTAC [11:55:30]
My question pertains to the labeling. Was any consideration given to adding off target– possible off target effects to the label?
While I recognize that your preclinical studies did not show evidence of that, I think given the nature of this meeting and the emerging field and the difficulty of potentially trying to identify these things ahead of time, might that be something to consider?
Stephanie Krogmeier, Vertex [11:55:55]
Yeah, we are still in discussions with the agency on the label. We– those discussions will certainly include the safety of exa-cel.
…
Bill Hobbs, Vertex [12:04:46]
The first part of your question is about the number of cells infused in patients, and the protocol specified a minimum of 3 times 10 to the 6 per kilogram and a maximum of 20 times 10 to the 6 per kilogram.
And in the clinical study, that range was infused into patients.
…
Karl Kasamon, FDA [01:52:37]
The manufacture of Exa-cel, as shown in this diagram, starts with collection of autologous hematopoietic stem cells using apheresis.
Then, CD34+ positive cells are isolated, purified, and genome edited with a CRISPR endonuclease and an SPY101 guide RNA.
Following editing, the cells are incubated in a culture medium, washed and cryopreserved.
After completion of lot release testing and manufacture of the appropriate dose, Exa-cel lots are shipped to qualified clinical centers for infusion.
…
Karl Kasamon, FDA [02:00:25]
Exa-cel administration to patients with severe sickle cell disease led to achievement of primary and secondary efficacy endpoint by large majority of patients.
The long term outcome following CRISPR based gene therapy in humans remains unknown, and questions still remain regarding off target unintended genome editing.
…
Komudi Singh, FDA [02:03:09]
A growing body of evidence has shown that Cas9 mediated edits can also occur when there is an imperfect base pairing between the guide RNA and the genomic DNA in other locations, giving rise to unintended off target editing, as shown in the figure on the lower right portion of the slide. Next slide, please.
If an unintended genome editing occurs at the region of the genome known to have regulatory elements, then a double strand break in such locations can disrupt regulatory function. Similarly, off target editing at gene coding region can lead to gene inactivation.
If the gene happens to play an essential role in cell function, then such unintended edits can be deleterious. These edits can also increase the risk of cancer.
…
Komudi Singh, FDA [02:31:51]
The applicant performed off target safety assessment accounting for heterogeneity using the Thousand Genomes Project database.
However, the small number of sequencing data present in the database, and lack of confirmatory testing of all off target loci in samples harboring the variants is concerning.
We are also concerned about the adequacy of small sample size in cellular off target analysis with exa-cel and if the use of small number of healthy donor and sickle cell donor cells would adequately inform us of potential off target editing risk of exa-cel.
…
Gil Wolfe, CTGTAC [03:16:13]
It’s really exciting to see how many patients have been treated and how positive the results have been.
I think the other thing that I would mention with regards to off target analysis is that, we want to be careful to not let the perfect be the enemy of the good, and right now, I feel that you can do a lot of in-depth analysis with regards to cellular analysis, in silico analysis, and samples that are treated prior to introduction into patients and you want to do as good a job as you possibly can, but at some point, you have to just try things out in patients.
And, I think in this case that there’s a huge unmet need for individuals with sickle cell disease. And, it’s important we think about how we can advance therapies that could potentially help them.
And I certainly think that this is one of them.
…
Gil Wolfe, CTGTAC [03:18:51]
I honestly think that the number of samples that they’ve analyzed is reasonable.
There’s only so much you can learn from additional samples, unless you’re going to focus in on in my mind sequence variants and trying to find samples that would have sequence variants that would allow you to interrogate off target sites that aren’t common within the human population.
…
Alexis Komor, CTGTA [03:21:09]
The perfect off target analysis would be sequence the patient, use that as a reference genome, and then individually validate every single off target. And is that reasonable here?
Especially, I love the quote from Dr. Wolfe, expecting perfection at the expense of progress here? Like, do we have the technology to do that, to sequence every single patient and do an expansive individualized off target analysis on each one? Probably, but is that reasonable to expect from them at this point? I don’t know.
And then additionally, yeah, for GUIDE-seq, GUIDE-seq is a very specialized technique. It’s difficult to do in certain cell types. It nominates putative off targets. Many of those nominated off targets don’t end up being bona fide off targets just because they’re very, very, very low levels and the cell can repair those perfectly under genome editing conditions.
And so again, would the ideal analysis be to perform GUIDE-seq in the patient samples and then go in and individually validate each one? Yeah. But is that reasonable to ask? I don’t think so.
I think what we see here, I think, especially given the benefits of this of this treatment, of this cure, and what these patients are dealing with without having this treatment, I think the benefits far outweigh the risks here.
…
Lisa Lee, CTGTAC [03:25:06]
If we anticipated or there was some theoretical possibility that if this found just the right off target, somebody would drop dead, that’s a very different kind of risk than, I mean, even a leukemia, depending though there are lots of different kinds and they can vary with respect to lethality, etcetera, but given what people are dealing with right now, and given that the evidence for the efficacy of this treatment is overwhelming, I really wonder what would we not be able to tolerate with respect to the unknown.
…
Nicole Verdun, FDA [03:33:31]
We just heard a comment considering too much risk or outweighing benefit. And that was sort of not the setting that we were talking about this. So I just wanted to sort of make that clear.
We’re not here discussing any concern with the benefit, but we were more wanting to have a conversation about is whether the committee recommends any additional studies and just realizing that we also have certain regulatory authorities where those could be in the postmarket setting, so postmarket requirements or commitments or otherwise if needed.
…
Gil Wolfe, CTGTAC [03:35:09]
The one thing that I, I thought was missing that I I’d love to see is a molecular analysis of on target edits, the distribution of sequences, I think that’s what Dr. Bauer was getting at in his presentation that you can use those as sort of a fingerprint to look for clonal expansion potentially within the patient’s hematopoietic cells.
And, it seems to me that that sort of analysis would be relatively straightforward in principle. The applicant is already generating this data because they’re following the indel rates over time. So they’re actually sequencing peripheral blood to look at this.
And so it should be relatively straightforward to follow up with regards to the indel spectrum. And does it change over time, and would that provide a surrogate and sort of an early warning sign of something going wrong with regards to the hematopoietic system?
…
Gil Wolfe, CTGTAC [03:46:33]
I just wanted to follow up on the suggestion of whole genome sequencing.
So I think the challenge there is that editing rates at off target sites may be quite low. And so whole genome sequencing is great for getting sort of the sequence of the most common genome that’s present in an individual, but with regards to picking up low frequency edits, I don’t know that whole genome sequencing will be real effective for doing that.
I think it might turn out to be challenging. I think that the error rate might start to get at the point where it would be challenging, unless there’s a high rate of editing.
So, others may have more experience with whole gene genome sequencing than myself, but I’m not sure that it will give us the information that we’d like.
…
Alex Komor, CTGTAC [03:47:31]
I agree. I think the whole genome sequencing would be able to get like a reference genome for that particular patient, but, I don’t know, like the sequencing depth required to identify some of these low efficiency off target events, potentially.
I don’t know if that would work, but for identifying additional putative off targets and especially the one that we’ve been talking about all day, you could identify that.
…
Taby Ahsan, CTGTAC (Chair) [03:54:14]
So let me try to, uh, summarize a lot of the comments that were made.
Starting at the highest level.
One of the questions were, where are we on this risk mitigation curve? Are we at a point where we have the technology in order to really address these questions?
It does lead us to this thought that we have this theoretical analysis that can be done against reference samples or specific cells, but the safety aspect is really related on a per patient basis or a target population.
So one of the question becomes when have we done enough theoretical analysis to allow us to move forward? And that’s, I think the major question, that we want to look at.
I think overall the sentiment was that the in silico analysis was quite detailed. It used quite lenient thresholds so that the criteria were set to really be able to create a good list of off targets. Maybe there is some room there for doing some deeper sequencing.
There was also this GUIDE-seq empirical analysis that seems to be a growing standard in the field. It was appreciated that they were doing that, and it gave us different results from the in silico. And there were reasons behind that that seemed very rational that were presented by the experts.
And it just gives you more nominations to consider at the end of the day, there needs to be some assessment as to whether these off targets are biologically meaningful, and there might be some flexibility there in terms of how you evaluate that.
In terms of suggesting studies moving forward, there was quite a bit of discussion about the monitoring of the samples over the next 15 years. It would be nice to see some evaluation of monitoring the edits over real time, looking at Al expansion. But it’s unsure the technology that would be used to do that, whether whole genome sequencing would actually have the detection levels, to give us meaningful information there.
But thinking about new technologies related to long range sequencing would be very good, potentially monitoring the CPS1 variant.
But again, I think what it comes down to was that there was a robust approach using multiple methods to try to identify these off targets. And I think there’s opportunity to generate more data monitoring these patients moving forward.
There seems to be a deep a plan for a deep monitoring over 15 years. And that can be very, very helpful in generating data and monitoring these patients.
…
Gil Wolfe, CTGTAC [03:58:11]
I guess the only other thing that maybe we didn’t touch on, and I apologize for not mentioning this, but whether there would be a plan for pre-screening for patients that have a variant at CPS1 in the future and how that would affect whether they would receive treatment.
…
Taby Ahsan, CTGTAC (Chair) [03:58:41]
I think we talked about a lot of different aspects. There was a kind of a robust conversation that hopefully will be informative to the FDA as they start to evaluate different paradigms for off target analysis.
This is likely just the first of many more discussions around this topic as products come for regulatory approval.
ACIP discussion on influenza vaccines and ischemic stroke
- Source: webcast by Centers for Disease Control and Prevention
- Date: broadcasted October 25, 2023
Tom Shimabukuro, CDC [01:10:28]
There were no unusual or unexpected reporting patterns observed and no evidence of a safety concern detected for ischemic stroke with either of the bivalent mRNA COVID 19 vaccines in VAERS monitoring.
FDA monitoring in the CMS data and VA monitoring in the VA system did not detect any safety signals for ischemic stroke following bivalent mRNA COVID 19 vaccination using historical comparator designs.
A separate ad hoc CDC analysis during the bivalent Pfizer ischemic stroke signal assessment did not detect an elevated risk for ischemic stroke after influenza vaccination alone.
Surveillance conducted by international regulatory and public health partners did not detect a safety concern for ischemic stroke following bivalent mRNA COVID 19 vaccination.
There was no evidence of a safety signal for ischemic stroke detected in the manufacturer’s global monitoring of bivalent mRNA COVID 19 vaccination.
And no safety signals were detected for ischemic stroke for primary series or monovalent boosters for Pfizer or Moderna vaccines in U.S. and global monitoring.
And data suggests COVID 19 and influenza disease are associated with an increased risk of stroke. And this was previously presented at an ACIP meeting. Next slide.
So I want to show some data on disease because I think it’s– I think it’s important, in the context of these findings.
So this is a graph that’s actually taken from the FDA study, that I summarized previously.
This is monthly incidence rates of non hemorrhagic stroke in Medicare claims data, which suggests seasonality. And there are other data that actually show this pattern of seasonality. But this is fairly recent data.
And as you can see, incidence tends to be higher in the winter months, then dips down in the summer months, and then starts creeping back up in the fall.
And like I said, this pattern been observed in other analyses, and this is some indirect evidence of a possible correlation with circulating viral respiratory pathogens.
…
Tom Shimabukuro, CDC [01:14:22]
Just to provide some context for why I’m showing this.
There is some evidence that infection is associated with stroke, particularly with COVID 19 and flu.
So I think– we think it’s important to also consider respiratory illness and viral illness when looking at these vaccine safety data, next slide.
…
Tom Shimabukuro, CDC [01:14:53]
Our interpretation of these, of the data that I just presented from these post signal analyses after the initial RCA finding was there’s variable and inconsistent results that were obtained in some analyses of the risk of ischemic stroke, following bivalent COVID 19 vaccination, simultaneous bivalent COVID 19 and influenza vaccination, and influenza vaccination alone.
There is a lack of consistency in findings from different data systems when using different methods across age groups and across subgroup AEs.
The most common findings across the studies are finding of no association.
Multiple comparisons were conducted in studies while adjusting for multiplicity. And few of these reached statistical significance.
The studies were not designed to account for the potential protective effect of vaccination on stroke and later post vaccination periods.
Adjusting for seasonality and restricting analyses to chart verified cases frequently resulted in attenuated findings or findings that were no longer statistically significant.
The remaining statistically significant findings tended to be relatively small in magnitude and ischemic stroke cases in the analyses are predominantly occurring in older people and in people in the upper ranges of the age group studied, meaning there are relatively few cases in younger people. Next slide.
…
Tom Shimabukuro, CDC [01:16:21]
So the available data do not provide clear and consistent evidence of a safety problem for ischemic stroke with bivalent mRNA COVID 19 vaccines when given alone or given simultaneously with influenza vaccines or when influenza vaccine is given alone.
Most study results have not shown an association between vaccination and ischemic stroke and no clear pattern demonstrating increased risk has emerged.
Seasonality of stroke risks and an unusual respiratory illness pattern in 2022 and 2023 could be impacting the results of some of these analyses and unrecognized SARS-CoV-2 infection could also play a role in occurrence of stroke after vaccination.
…
Tom Shimabukuro, CDC [01:17:45]
So our next steps will conduct additional analyses on the possible relationship between ischemic stroke and bivalent mRNA COVID-19 vaccination, simultaneous administration of bivalent mRNA COVID 19 and influenza vaccines and influenza vaccine alone.
We will continue vigilant safety monitoring of the 2023-2024 COVID 19 and influenza vaccines, including for ischemic stroke.
And we will be happy to come back to the committee and brief you when we have additional safety data.
…
Jamie Loehr, ACIP [01:18:29]
I’d also like to say thank you very much for that presentation. Could we go to slide seven?
A little bit of context, the flu work group heard about the FDA study, which is on slide seven here, and there were some concerning findings. And so that was about two weeks ago last Monday.
So two days ago, the presenter gave us a little bit more detail about this, and the work group asked for a context and a presentation today.
So this all came together from Dr. Shimabukuro in 48 hours. And I really appreciate that detail and perspective of all the studies, not just the one study that came out two weeks ago.
So I feel like I have a much better context of the vast number of studies, which are suggesting that there is not a consistent pattern of concern.
…
Tom Shimabukuro, CDC [01:22:01]
We have multiple complimentary systems that work in different ways and use different methods, both within CDC and across the federal government, with our partners, such as FDA, VA, and Department of Defense.
And our systems are, as you said, designed to be sensitive, to be able to rapidly detect and assess potential safety problems. And we strive to be timely and transparent in our communication of safety information.
And I agree with you that consistency of findings across different systems using different methods is important and tells us something about a potential adverse event.
And the myocarditis signal is a great example of where all the evidence from multiple systems in the United States and outside were all pointing to a common finding.
And I think this– this is a much different situation than myocarditis.
October 30, 2023
ACIP discussion on pentavalent meningococcal vaccine wording
- Source: webcast by Centers for Disease Control and Prevention
- Date: broadcasted October 26, 2023
Katherine Poehling, ACIP [01:14:58]
The second one is on slide 51, about the pentavalent meningococcal vaccine, and it’s about the wording.
Patricia Wodi, CDC [01:15:08]
Oh, sorry.
Katherine Poehling, ACIP [01:15:09]
Okay. No problem. Oh, maybe I’ve got the wrong number. I apologize.
Patricia Wodi, CDC [01:15:13]
This one?
Katherine Poehling, ACIP [01:15:25]
If you go back to the previous one– wait– uh, yes. This one.
I got– I was– I’m wondering about the wording because at the end, we talk about how you can have, when both of them are going to be administered on the same day in a single injection with Pen-actra is preferred.
It kind of sounds like we prefer that and we did not make a preferential recommendation. And so I want– I worry that this would be misinterpreted.
Patricia Wodi, CDC [01:15:57]
Yeah, we went back and forth with if indicated, what we wanted to– we wanted to communicate that if the patient or the provider had a preference, but we weren’t sure how to word that.
Katherine Poehling, ACIP [01:16:16]
I think that it’s okay, because you said that they may receive a single dose as an alternative to separate administrations, when both would be given in the same day.
And I would add a period, because I think that clearly communicates that both are equally reasonable options.
Patricia Wodi, CDC [01:16:36]
Okay, we can do that.
Katherine Poehling, ACIP [01:16:36]
Okay, and yay.
All right, and then, PCV 15 and 20.
Okay. No, this is going to be okay. Alright. Stop.
Patricia Wodi, CDC [01:16:57]
Okay.
Katherine Poehling, ACIP [01:16:57]
Back up.
Patricia Wodi, CDC [01:16:58]
Back up.
Katherine Poehling, ACIP [01:16:59]
The routine. All right, so what I’m worried– I do appreciate the routine and special, but what I’m worried about is that people will look at the routine and not realize because we’ve changed the recommendations for the special.
And so, I like this sentence or, but it kind of says that if you’ve received the complete four dose series you’re done, unless you have a condition listed in special situations, and I’m thinking you want to add that so people look at the next page and that is the end of my suggestions, and only three is really good.
Grace Lee, ACIP [01:17:50]
Excellent. Thank you, Dr. Poehling. Ms. Bahta?
Lynn Bahta, ACIP [01:17:54]
Dr. Poehling, you always beat me to the hand raising.
I also had similar, um, concerns about the language of preferred and actually went to the thesaurus and looked at desired or chosen, but even, I don’t even think it’s a important part of the sentence.
It doesn’t need to be said.
And then I know that we do not make policy with this schedule, but I wanted to raise an issue that seems to be kind of bubbling up related to HPV and giving HPV in a more routine recommendation starting at nine, rather than– well, it can be given as early as nine.
So, that’s just a– put it on the parking lot for an HPV work group. Okay.
The question is– there’s data that growing data that shows better completion of HPV when you start the series at age nine, it kind of removes some of the sexual implications, and there’s– it’s easier to talk about cancer prevention.
But I know that the data is conflicting to some degree, but I just am hoping that the work group could look at that more closely.
Melinda Wharton, CDC [01:19:26]
So, I know that there is interest in reconvening the HPV work group to address– I believe this issue is as well as probably some others.
Given the large amount of– the large number of work groups the committee has currently had to support, we’ve been delayed on getting that started, but hopefully we’ll be able to do that soon.
ACIP discussion on RSV immunizations for pregnancy and babies
- Source: webcast by Centers for Disease Control and Prevention
- Date: broadcasted October 26, 2023
Julian Ritchey, Sanofi [00:37:05]
On behalf of Sanofi vaccines and our alliance partner, AstraZeneca, appreciate the opportunity that NCIRD is providing for us to comment on the current market situation for Beyfortus or nirsevimab.
As Dr. Daskalakis has shared, we’re experiencing unprecedented demand for Beyfortus in response to the unmet need that has existed in RSV prevention.
Despite an aggressive supply plan built with the goal of outperforming past pediatric immunization launches and built to anticipate the demand of the season, the demand that has materialized has been much higher than forecasted across both the 50 milligram and 100 milligram presentations.
Both product dose presentations do continue to ship to fulfill existing orders. However, for the 100 milligram presentation, we have stopped accepting new orders as demand has consumed the supply currently available for this season.
We will continue shipping doses of the 100 milligram to fulfill orders already on hand, over the coming weeks.
Orders are still being accepted for the 50 milligram presentation.
We’re working closely in collaboration with the Centers for Disease Control and Prevention to ensure equitable distribution of available doses through the VFC program.
Our approach for fulfilling existing orders and in taking new orders for the 50 milligram product across the private marketplace will also be done in a similar manner with equity in mind.
We appreciate the clinical guidance provided by CDC earlier this week via the HAN that was mentioned before. Additionally, we’re working with the FDA together with AstraZeneca, our partner in charge of the manufacturing and supply, to deliver all of the doses planned for this season.
We appreciate the challenge that these supply constraints present for providers and parents, as well as for CDC, the FDA and AAP and others.
As we introduce Beyfortus, we are thankful for your patients and your collaboration.
We’re already working to ensure there will be sufficient supply available for next season as we continue to focus on making this season’s doses available as rapidly as possible.
…
Demetre Daskalakis, CDC [00:40:19]
We will have sort of a better view of what’s happening in terms of coverage as we go further in the season, but in general, I’ll say that because this is a commercially distributed vaccine, we’ve been really in close contact with manufacturers and distributors to have a sense of sort of what’s happening on the ground as well as really close discussions with our [stakeholders] around the country, including professional organizations, etcetera.
So I’ll start by saying it does– there does not appear to be a bottleneck in production of the vaccine. So, from the perspective of what we’ve heard from manufacturers, as well as from distributors, vaccine is flowing.
What we are hearing is that there are some barriers related to concerns around coverage. So CDC is actively engaging across [stakeholders] that deal with coverage, both governmental, CMS, and also others to make sure that the message is clear the importance of this vaccine product for pregnant people and for their newborns.
Additionally, many engagements have happened and more are planned to be able to sort of tout the importance of the vaccine and also to do some myth busting around sort of what is available as well as what we sort of forecast will be the coverage scenarios.
There will be some coverage issues because there are some limits to the speed at which a private insurer needs to cover this, but we are engaging with them as well to, again, highlight the importance of a public health view into strategies that tend to be more on the commercial side of the fence.
…
Sarah Long, ACIP [00:42:05]
We should be winding down on need for the 100 milligram vials as we catch people who are a little older at the beginning of the season in the next month, but the 50s we’ll need through March and they are the most vulnerable.
Do you– at what level of uptake did you plan for and what do you anticipate as far as potential shortages of the 50s?
Julian Ritchey, Sanofi [00:42:37]
Thank you, Dr. Long, and as far as the 50s, I don’t have specific numbers to share in terms of volume, but it’s something we’re watching very closely.
At this point, seeing the level of ordering that happened early on, we’re still trying to understand how much of that ordering is early in stocking ordering, and the utilization that will go forward.
So, the CDC can speak to more of the specifics of the VFC program details, but for non VFC and private sector doses, we’re monitoring that 50 milligram order very closely and will work to adjust any order quantities going forward if we detect that.
CDC officials at ACIP meeting on COVID-19 vaccines
- Source: webcast by Centers for Disease Control and Prevention
- Date: broadcasted October 26, 2023
Shannon Stokley, CDC [00:11:36]
We monitor vaccine doses for the program both through our pharmacy partners and our immunization program partners.
The contracted pharmacies are required to provide to us on a daily basis the number of sites that are operating and how many doses have been administered. And then for our public health partners who receive COVID vaccine doses through CDC, we’re able to track the orders, including where they go, the number of providers ordering vaccine and the number of doses that were ordered.
So currently there are over 24,000 contracted pharmacy locations and over 380,000 doses have been administered by these sites. And there are now over 4,400 public health safety net providers that have placed more than 6,100 orders for over 287,000 doses of COVID 19 vaccine, using their 317 funds.
And nationally 81% of people without insurance are estimated to live within a five mile drive distance to a Bridge Access program provider.
…
Shannon Stokley, CDC [00:13:25]
Today I’m just gonna provide some preliminary data about vaccination coverage. This is from our National Immunization Survey. We have an adult COVID module and a child COVID module that’s administered every week.
For those not familiar with the NIS, this is a random digital telephone survey of cell phone numbers. And vaccination status is self-reported by the respondent. And, just to caution that sometimes there may be the potential for over estimation since it’s self reported.
But looking at our data for the week of October 8th through the 14th, and for adults 18 and over, we see that an estimated 7% are reported to have received a COVID 19 vaccine. And this is the 2023-2024 vaccine.
We also assess vaccine intentions among individuals who have not received a vaccine. And we’re seen about 25% of adults say they will definitely get vaccinated, and about 38% saying they probably or definitely will not get vaccinated.
…
Shannon Stokley, CDC [00:14:51]
COVID 19 vaccine was more frequently reported among adults who were older, had health insurance and had higher incomes.
…
Shannon Stokley, CDC [00:15:07]
This is children through age 17 years. Again, the time period is October 8th through 14th, and this is parents reporting the vaccination status of their children.
And we see that about 2% of children were reported to have received a COVID vaccine. Next slide.
…
Shannon Stokley, CDC [00:16:13]
We show that a little over a quarter of children have a parent that said they definitely will get their child vaccinated, excuse me– and about 38% have a parent that said they definitely will not get vaccinated.
…
Sarah Meyer, CDC [00:21:08]
From what we’re hearing, it’s not necessarily a supply issue. It’s not necessarily a distribution issue. Although, of course, as alluded to there have been some things that have had to be worked out through commercialization.
I think early on in the rollout, there was a little bit of a delay in getting the Moderna supply, but that has improved.
I think what– what we’re hearing about more and more is just how difficult it is for pediatricians to be able to carry these products, to store them, to stock them, just the kind of implementation issues around providing this vaccine.
And I think we do have some good updates, which I know, Dr. Wallace will be talking about in terms of some of the new guidance that we hope makes it more implementable in the pediatric office.
But we are hearing about some of those issues from the VFC side. We have been continuing to increase the allocations so that states can order more VFC doses. And we have recently published an addendum to our VFC operations guide that gives some additional flexibility around borrowing and things like that to help further smooth some of the implementation challenges that pediatric providers are facing.
…
Megan Wallace, CDC [00:38:15]
I want to end with providing a preview of upcoming policy discussions.
First on the horizon is the consideration of additional COVID 19 vaccine doses in older adults. We anticipate this discussion during the February 2024 ACIP meeting.
And if we aim to bring that before ACIP at that meeting, the policy discussion will occur prior to individuals reaching six months since their last dose.
Then we will pivot to preparations for future COVID 19 vaccine formula updates with those discussions beginning at the June 2024 ACIP meeting.
As always, we will be continuously monitoring vaccine effectiveness, vaccine safety, and COVID 19 epidemiology, and COVID 19 vaccine recommendations can be updated if needed.
October 27, 2023
HHS officials on commercialization of COVID-19 treatments
- Source: briefing with HHS officials
- Date: hosted October 27, 2023
HHS Official (1) [00:04:58]
But as I said, we will continue to have free federal product while this process is working itself and all those kinks get worked out.
HHS will continue the work of encouraging insurers and providers to update their system and encourage coverage for these drugs ahead of that transition, and will continue to work with providers, manufacturers, distributors, payers, and stakeholders on that orderly transition in the weeks and months ahead.
We’ve already convened calls, as we did the vaccine transition, we’ve already convened calls with the plans, the PBMs and the pharmacies to check in, and we’ll be doing that periodically as this transition goes.
…
HHS Official (1) [00:07:09]
We don’t always have that precise estimate because that’s sort of literally by definition out of our view in terms of what doses in there are in the field.
We do think that there are millions of doses in the field right now. We anticipate probably at least somewhere around two and a half million of Paxlovid and at least 1 million of Lagevrio.
But those ranges are not certain. And we’re not sure of what the top range of that estimate may be.
…
HHS Official (1) [00:07:42]
I want to be clear, we are encouraging all the payers to be working with those manufacturers to establish agreements and established contracts.
We do anticipate, and this is not USG anticipates, but our feedback in terms of conversations and what we’ve been told so far is that a lot of those agreements will get settled, for Pfizer. And some of those agreements will be settled for Lagevrio.
We’re keeping a close eye on those conversations and negotiations, although again, that’s not ours to work out and would direct you back to the companies and the manufacturers to get a better sense of that.
But the bottom line here for us and what we’re sort of this plan right now we’re talking about is because there is still that government product in the field, we are allowing time for some of those kinks to get work through. We are allowing time for those agreements to get settled and all those transactions to figure out and clarity most importantly, for providers and patients, in terms of how they can access that treatment through those commercial channels.
And in the meantime, we are leaning forward to use what product we have to ensure that there is a safety net to be sure that process can go smoothly and has the time it needs that we understood and learned and we are able to do that in this case in a way that we were not able to do in the vaccines to provide this overlap period and ensure that there was adequate time for that the assistance to stand up and be effective.
…
HHS Official (1) [00:11:12]
All of our courses in currently, and in the field, and in federal supply, were due to expire starting as early as February of next year and every dose in the field or under USG possession would have expired by summer of next year.
So what we have worked out in terms of, with this deal, in terms of protecting Medicare, Medicaid, uninsured, all the way through 2024, and ensured a stockpile beyond that 2024, for specific uses like emergencies or uninsured, that is all benefits that we did not have under current deal.
So I want to be clear about sort of where our current world was in terms of why we had this deal with Pfizer, and we were so interested in making this deal, that expiry was looming.
…
HHS Official (1) [00:15:38]
That’s partly a lesson from the vaccines. It’s also an option we didn’t have for vaccines, because that was a product switchover, right? We had one product EUA-authorized and the commercial product was authorized. There was no ability to have any overlap, because of, it was simply two different products.
These are essentially the same product, although they may be labeled EUA versus commercial product, it’s the same drug entity.
Why does that matter? What that means is that we were able, in a way that we weren’t with vaccines, to create this overlap period that’s going to begin on November 1.
…
HHS Official (1) [00:16:12]
So November 1, commercial channels begin on November 1, but I don’t think many people are going to notice because we have all that federal product in the field.
So I wouldn’t say it is one of the lessons, but it was a lesson we weren’t able to do in vaccines. It wasn’t like we didn’t want to do that then.
But what we are able to do because of that overlap is building that ensure that there’s ample supply out there.
…
HHS Official (1) [00:17:24]
November 1 is not gonna be a landmark day, but something that you know is mostly worked out between the plans, providers, in terms of systems, getting those systems worked out, and giving them a chance to work on all those kinks before anyone has to rely on it.
And once those systems are up and running, once a pharmacy says, ‘okay, our systems are working, we tested them, we’re ready to go,’ then at that point, they can say, ‘okay, we’re ready to make that switch. Let’s return our EUA product back to USG, let them get the credit forward.’
They can make that choice when they’re ready to make that transition.
…
Question [00:18:17]
Does that mean that at some point during this transition, there’s going to be a point where like, if I’m a patient, I’m looking for Paxlovid, my insurer doesn’t yet cover it, that I’m going to have trouble finding a location that’s still offering federal supply?
And then I have a follow up to that.
HHS Official (1) [00:18:36]
I think we’re encouraging the flexibility for the providers to have that ability to make that decision, as we said, we’re going to have plenty of product out there.
We would generally hope that the commercial insurance has straightened out by the time any pharmacy says we’re ready to make that flip.
And again, once they make that flip, as I was saying before, because of the deals we’ve negotiated, particularly with Pfizer, a lot of those products will be available even after that commercial transition, at no cost for people on Medicare and Medicaid and without insurance.
And again, for commercial because of if the coverage is there, and we hope that they can work out those deals before that transition, in addition to the coverage, there’ll be that co-pay program that Pfizer has committed to.
…
HHS Official (1) [00:19:19]
So I guess my point is that, while each individual may have a different route to get low or no cost access, our real strong hope, and we’re going to see how this goes and we’re gonna be keeping close tabs and conversations with everybody is that, you have free final product for now.
Once you make that flip, you can use the access program. You’re ready to go with insurance coverage, you’re ready to go with all the various other programs to ensure that the access remains post swap.
…
Question [00:19:48]
You had talked earlier about kind of the expiry of these EUA labeled courses. Is that what decided the timing of this transition?
I guess I’m just trying to understand why it couldn’t wait until after the winter COVID wave, and then after you’re sure that all the insurers are covering the EUA label– or the NDA labeled courses? Talk about the timing of this transition. Thanks.
HHS Official (1) [00:20:09]
As I said, we have product that’s going to expire as early as February. We know that, and so that was driving some of the conversation.
This is also part of the negotiated deal with Pfizer in particular, to ensure that they could enter the market smoothly.
In the meantime, we were able to, as I said before, preserve the USG value and no expired doses, preserve the access for Medicare and Medicaid, and get a stockpile out of that deal as well.
So this was part of the negotiated deal with Pfizer in terms of the timing, but yeah, we were gonna have doses starting to lose as soon as– we weren’t going to be able to get through this winter surge without doses expiring, if we didn’t come to some other arrangement.
…
Question [00:23:46]
Pfizer said that they will book revenue as they send updated Paxlovid to the government. Is the government paying anything more than what it is already paid? And I have a follow up after that.
HHS Official (1) [00:23:58]
[HHS Official] check me, but I believe the answer’s no?
HHS Official (2) [00:24:02]
That’s correct.
…
HHS Official (1) [00:24:56]
Where we were sitting when we entered that table with Pfizer, all of our courses were due to expire, right? So we were going to walk away going into next winter surge with nothing, no Medicare, no Medicaid, no uninsured, no dose in federal supply, nothing for emergencies.
So with this deal, we have don’t lose no dose to expiry. We protect Medicare and Medicaid insured through all of 2024.
And we get a stockpile beyond that through 2028 for specific uses of uninsured or emergencies. So we will continue to look for places wherever we can to make the fight and fight the fight for pricing. And I think we’re trying to prioritize and understand where we can get in any particular deal.
CDC’s figures on nursing home residents up to date on COVID-19 vaccines
- Source: dashboard by Centers for Disease Control and Prevention
- Date: accessed October 27, 2023
- DC 25.8%
- SD 23.5%
- ND 16.0%
- NM 15.6%
- RI 15.1%
- NH 14.7%
- AK 14.0%
- MT 13.3%
- VT 12.7%
- OR 12.1%
- FL 11.9%
- WI 11.7%
- NJ 10.9%
- PA 10.3%
- NV 10.2%
- CO 10.1%
- MN 10.1%
CDC officials at clinicial outreach call about respiratory viruses
- Source: webinar by Centers for Disease Control and Prevention
- Date: broadcasted October 26, 2023
Sarah Meyer, CDC [00:25:18]
Ordering of RSV immunizations is very similar to how other immunizations are ordered. So essentially through routine mechanisms, for example, directly from the manufacturer or wholesaler or distributor.
For Vaccines for Children providers, these immunizations are ordered through the state or local immunization program. Again, very similar to how this occurs with other routine immunizations.
…
Sarah Meyer, CDC [00:26:58]
For nirsevimab, this is priced at about $495 per dose, using the private sector costs.
There are some payment flexibility this season. So the manufacturer has stated that there is 150 days for payment when ordering directly from the manufacturer.
And in terms of insurance coverage, this immunization is covered under the Vaccines for Children or VFC program.
And similar to the maternal RSV vaccine, most insurance plans are required to cover this product, but do have one year to do so.
…
Sarah Meyer, CDC [00:43:44]
We did recently publish an addendum to our operations guide, which is now also published online, that provides some additional flexibilities or considerations for nirsevimab, particularly for this season.
We do have a ramp up period for private inventory requirements.
And although VFC eligible patients remain the priority for VFC doses, we do have some flexibility around bidirectional borrowing between private and public stock allowed in certain situations and where allowed by the jurisdictional policies.
One point I will just make though, in the setting of supply constraints, it could be difficult to implement borrowing. So this was– this is really a flexibility meant for those practices that do have sufficient private and public stock to be able to to do borrowing.
…
Sarah Meyer, CDC [00:46:02]
What I really wanted to highlight is that there is some increased flexibility for interchangeability of COVID 19 vaccines.
We have heard the feedback that strict homologous dosing for these vaccine products is a barrier for pediatric practices, especially for those who carry– who are only able to carry one of the vaccine products and not all of them.
So we do have some increased flexibility around mix and match, for those pediatric doses that require more than one dose in the series.
So the previous language around this was that in the following exceptional situations, a different age appropriate COVID 19 vaccine may be administered under, the following conditions, which are listed there. So a very pretty strict language around when it’s permissible to give a different vaccine.
So for example, if somebody got Moderna for their first dose, very strict language about them meeting Moderna for the next dose, except for exceptional situations.
The language now is more flexible. And we say in the following circumstances, an age appropriate COVID 19 vaccine from a different manufacturer may be administered when the same vaccine is not available at the vaccination site at the time of the clinic visit, the previous dose is unknown, the person would otherwise not receive a recommended vaccine, or the person starts, but is unable to complete, a vaccination series with the same COVID 19 vaccine due to a contraindication.
So we hope that this increased makes it a little bit easier for our pediatric providers to give COVID 19 vaccines to this youngest age group.
…
Jefferson Jones, CDC [00:51:40]
So if there’s no nirsevimab available, if they’re palivizumab eligible, as in they’re high risk infant per AAP’s recommendations to receive palivizumab, palivizumab is eligible.
If they’re not eligible for that, unfortunately, there’s nirsevimab particularly, there may be– we’re suggesting using available doses for those at the highest risk.
For others, there’s every day preventive measures that can limit such as trying to have household contacts wash your hands and avoid touching face, avoiding close contact with sick people, covering cough and sneeze, etcetera. Those everyday preventive measures are something that that can be tried, but there’s no other specific immunizations available.
…
Jefferson Jones, CDC [00:55:14]
For this season, in particular, I think that there may be a lack of nirsevimab availability, and to forego maternal vaccination and then not have nirsevimab available after birth would be quite unfortunate.
So if, unless you know that nirsevimab is available, that is something to strongly consider.
Efficacy trials comparing the two products head to head have not been conducted. And, although the point estimates differ, the trials have different definitions of endpoints and are difficult to compare.
The antibody data suggests that nirsevimab efficacy may last longer with a longer half life than the maternal vaccine.
But for the many people, for these infants being born during the season, we expect that protection will last through the season for either of the products or for at least the majority of the season.
So, it can be patient preferences avoiding an injection in the infant. It might be any concerns of– some people are more concerned about being vaccinated during pregnancy than others.
So I think there’ll be a lot of individual patient preferences to consider.
October 26, 2023
Abbott spokesperson on FDA warning letter about Similac Probiotic Tri-Blend
- Source: email from Abbott spokesperson
- Date: received October 26, 2023
This issue pertains to a single probiotic additive for formula called Similac Probiotic Tri-Blend, which is used by fewer than 200 hospitals. Importantly, this does not apply to any of Abbott’s infant formula products available at retail, and parents and caregivers can continue to find and use Similac infant formulas nationwide. This Warning Letter is not related to any manufacturing quality issue. This product has been used for the past several years and has had a strong safety profile.
This product has about $1 million in sales.
October 25, 2023
ACIP meeting discussions on mpox vaccines
- Source: livestream by Centers for Disease Control and Prevention
- Date: broadcasted October 25, 2023
Pablo Sanchez, ACIP [00:11:31]
So the potential implications of the interim routine recommendation that we plan, that will be for vote today, will increase vaccine coverage and prevent or minimize future outbreaks and control the current outbreak.
It would remove some stigma and facilitate one-to-one consultation with clinicians during their appointments for vaccination, next slide.
And also, it heralds the potential commercialization of the Jynneos vaccine.
The product sponsor Bavarian Nordic has indicated that they will attempt to commercialize the vaccine if it is on a routine schedule. And so this will transition the vaccine from U.S. government’s stockpiles, which are intended for smallpox preparedness, to the commercial sector.
…
Pablo Sanchez, ACIP [00:12:28]
So this is the tentative timeline for ACIP discussions and votes.
So today would be interim routine recommendations and clinical guidance. Then in early of 2024, publication of two MMWRs are planned. The use of the Jynneos vaccine during mpox outbreaks and the use of the Jynneos vaccine among persons at risk during the ongoing outbreak.
And possibly in 2024, we would consider the results from an ongoing NIH trial on the use of the Jynneos vaccine in adolescents aged 12 to 17 years of age.
And then to be determined would be the review of epidemiology, cost effectiveness analysis, and other data to determine if routine recommendation should be continued beyond the next two to three years.
Next slide.
So, again, the proposed recommendation would be the ACIP recommends vaccination with the two dose Jynneos vaccine series for persons aged 18 years and older at risk for mpox.
…
Faisal Minhaj, CDC [00:18:12]
We still see cases at an average of one to four cases per day, and have not been consistently below this level, highlighting that mpox has not been eliminated from the United States. Next slide.
When looking at the cases in 2023 geographically, we see that cases are occurring across the U.S. in different jurisdictions. This map highlights cases detected from January to October, from each jurisdiction.
Looking at the image, we can see that cases are spread out. Many of these are not linked to other known cases, suggestive of continued community transmission or potential underdiagnosis.
…
Faisal Minhaj, CDC [00:19:17]
We’ve also seen cases in patients following vaccination.
This has been reported since the outbreak started, including some clusters, but is relatively rare in comparison to the total case counts.
Importantly, most patients with infection following vaccination have mild illness.
On the right hand side of the screen, we highlight data from a recently published manuscript on infections following vaccination.
You can see that the median number of lesions in these patients was two with an inter-quartile range of one to five.
Additionally, only 2 out of 30 patients received tecovirimat, and few were hospitalized, suggestive that illness was mild following vaccination.
…
Faisal Minhaj, CDC [00:19:58]
Reinfection of mpox is rare and was not well reported until the current outbreak.
Potential reinfection cases have been published in the literature, but only a few with convincing evidence of true reinfection.
On the right is an image from a recently published case series where the authors found a total of eight probable cases of reinfection.
Reinfection appears to be mild with a lower lesion count and duration of rash.
CDC is aware of less than 10 cases of probable reinfection, which makes up less than 0.001% of cases.
…
Faisal Minhaj, CDC [00:28:27]
Models not only estimate that vaccination is key to ending the mpox outbreak, but also that vaccination is key to preventing mpox resurgence.
Later in the outbreak in D.C., the model estimated that nearly the entire high risk population of MSM who engage in one time sexual partnerships had gained full or partial immunity through vaccination or through acquiring and recovering from mpox, making resurgence in D.C. unlikely.
However, over time, population level immunity will decrease due to population turnover. Further, most U.S. jurisdictions had a lower vaccine coverage than D.C., potentially leaving them vulnerable to resurgence.
This plot shows the probability that five infectious individuals in a population of MSM would cause a resurgent mpox outbreak on the Y axis over various levels of immunity among individuals at increased risk for mpox on the X axis.
For the purposes of this model, either one or two dose vaccination is used. However, we know that from VE data, two doses is greater here.
We define that resurgent outbreak as continuous community transmission for at least three months.
We see that the risk of recurrence decreases linearly as population level immunity increases. Next slide.
While the probability of recurrence decreases linearly with population level immunity, the size of potential outbreaks has a more complex relationship with population level immunity.
Here the Y axis now shows the predictive cumulative cases if an mpox resurgence were to occur compared to the number of cumulative cases in 2022, over the population level immunity on the X axis. Next slide.
This model estimates that resurgent outbreaks will be very small, if the population level immunity is greater than 50%. Currently, only seven jurisdictions are above 50% of at least one dose Jynneos coverage among the high risk population. Next slide.
…
Faisal Minhaj, CDC [00:31:34]
Next we wanted to present updated VE estimates from the multi-jurisdictional case control study that was performed in 12 U.S. jurisdictions and initially presented in February.
Here cases were identified from a jurisdiction’s list of mpox cases while controls were identified from healthcare settings, providing HIV PrEP or STI clinics. VE here was adjusted for age, race, and ethnicity and immunocompromised immunocompromising conditions. It was stratified by route of administration and immunocompromised status. Next slide.
Overall VE from partial or one dose vaccination is updated here to 73% with a confidence interval of 59 to 82 with similar results for those with either administration route. This estimate is similar to the previously reported 75%, but does have a smaller confidence interval. Next slide.
Overall VE from two dose vaccination is updated to 83% with the confidence interval of 71 to 90, again, with similar results with either administration route. These updated data suggest that the VE estimates are stable. Next slide.
…
Faisal Minhaj, CDC [00:37:34]
There are cases following reinfection that CDC is currently investigating, some of whom have been vaccinated between their first and second infection. However, the number here are really small, and interpretation of this data is– can be challenging.
…
Agam Rao, CDC [00:39:00]
We have looked to see if there was any pattern, for example, with the Chicago cluster, where there was a cluster of cases that occurred in May that was affecting a lot of people who were fully vaccinated. And there does not seem to be an association with those breakthrough infections and the route of administration.
So we saw breakthrough infections that occurred among people who received two doses, intradermal, two doses subcutaneous, and also one dose subcutaneous and one dose intradermal. And, actually there were fewer that received two doses intradermal that were breakthrough cases from that cluster.
And I think the national data also mirrors that same thing.
…
Agam Rao, CDC [00:53:49]
The CDC multi-jurisdictional study is continuing to collect data. And, to our knowledge, is perhaps the only source of data that will be continued worldwide.
We’re attempting to estimate the VE preventing infections among immunocompetent versus immunocompromised persons, as Dr. McCollum has already mentioned, so hopefully we’ll have more information, but we certainly recognize that this is a major gap in our understanding.
…
Agam Rao, CDC [01:00:23]
When we went back and actually interviewed the patients who were impacted by that outbreak, 18 of those patients agreed to an interview and they were – they were people who were fully vaccinated, partially vaccinated and unvaccinated, and they actually indicated support for the vaccination.
So although chatter, I guess, online chatter indicated that there was some skepticism, the actual patients affected by that outbreak were supportive of the vaccine. Most stated that they would recommend vaccine to others. Some assumed that– some assumed that any mpox infection would’ve been prevented by the vaccine, but they still felt that the vaccine was effective in reducing the severity.
And unvaccinated persons said that they had not been vaccinated because they’d attempted to early on in the response, and then the supply was limited at the time, and then once they were able to, once it was available, they thought that since case counts had decreased, that the risk was very low and they didn’t end up seeking it.
…
Agam Rao, CDC [01:01:36]
Recently in June, so just a few months ago, CDC performed an online focus group from 52 people to help develop CDC communication material and participants were included if they identified as men were unvaccinated for mpox, never diagnosed with mpox, were 18 to 45 years of age, and had sex with two or more men within the past six months. So essentially people who were eligible for the mpox vaccine.
And participants were intentionally of diverse racial and ethnic backgrounds and were shown various communication materials and development by why CDC. Exposure to the information about mpox vaccine, safety, effectiveness, and hearing that there is a current threat of mpox affected their interests.
So although some of the individuals came to this focus group not interested in the vaccine or ambivalent about the vaccine, that changed, and many people disinterested in the vaccine became interested. They actually said that the current risk of mpox and protecting their community was motivating.
…
Agam Rao, CDC [01:08:41]
So Jynneos has actually been stockpiled for smallpox preparedness, but it was provided during this response, since it is not commercially available.
The doses would need to be replenished and it has been, and it will continue to be, a significant use of resources, for example, shipments and transportation personnel monitoring all of this during the peak of this outbreak.
And that would have to continue, and be a further drain on the SNS, the Strategic National Stockpile should this vote be passed, but in the future it’s possible that the vaccine will be commercialized. And if so, there are unknown costs that will be associated with that.
…
Agam Rao, CDC [01:12:56]
We’re uncertain if or when it would be commercialized. We have colleagues from Bavarian Nordic who will speak during the Q and A session and give a short statement, but if it is commercialized, it would be via Medicare or Medicaid and commercial plans without a copay, uninsured would be given it via Vaccines for Children program and then some uninsured and underinsured adults could have difficulty, but that would be similar to the difficulty that people have with other vaccines.
And it would be premature at this time to, without knowing if it’ll be commercialized to say that much more in the ETR presentation.
…
John Beigel, NIH [01:23:51]
That adolescent stage was fully enrolled in September of this year, which means the peak immunogenicity visits actually just happened a few days ago, a day 42 visit.
So that– it is designed to be a year follow up. So that puts us to October of 2024 for the full data set.
You know, we can discuss with ACIP and with the Bavarian Nordic about early looks, but to actually see the full dataset, it is that the– the visits will be October 2024, which means the data will be sometime after that.
…
Laura Quilter, CDC [01:28:47]
I think we were trying to strike a balance. I appreciate the comments on wanting to avoid stigma and wanting to simplify things for providers, but also wanting to really ensure that the vaccination is going to folks that are at the most risk for mpox exposure.
And so this was even tweaked from, I think, an even more complicated version first time around. And so I think it’s challenging to strike that balance.
…
Agam Rao, CDC [01:29:19]
As a modeling expert in the division of STD prevention at CDC, do you have any thoughts about just how much bigger the population would be if it was referring to all MSM or all people with HIV versus what it’s described as on this slide? But in case you do, do you have anything to add?
Ian Spicknall, CDC [01:29:39]
Yeah. In terms of a larger population, we would be going from roughly 2 million to 4 to 6 million people.
So we would be enlarging the scope greatly, and like what was, what has been said, to folks who may not benefit from this sort of vaccine for reasons you described a bit ago.
…
David Boucher, ASPR [01:38:49]
ASPR is supportive of the transition of Jynneos to commercial distribution, as the next correct step in ensuring continued access for those who wish to get vaccinated for mpox.
I want to say also we’re also very committed to ensuring a seamless transition, if this does go to commercial distribution.
We are not going to have gaps in availability of Jynneos, as we pass the baton onto our partners at Bavarian Nordic.
…
David Boucher, ASPR [01:39:25]
At that time we put 400,000 vials into the combined thresholds across 64 jurisdictions.
To date, 57,000, about 57,000 of those have been ordered. And that means we still have well over 300,000 vials available immediately to jurisdictions anytime they need to restock existing vaccination sites or supply new ones.
We’re going to keep this supply reserved for the jurisdictions, as we work with Bavarian Nordic, and HHS partners at CDC CMS, and several others over the coming months, if we do need to plan and execute a smooth handoff.
…
Lee Ann Kimak, Bavarian Nordic [01:41:40]
We believe that the routine preventative recommendation for at-risk populations under consideration by the committee is a critical step forward in addressing these inequity and access challenges.
This recommendation would allow providers and retail pharmacies to administer the vaccine where people are most comfortable seeking one, whether it’s at the pharmacy at their doctor’s office or at their neighborhood health clinic.
If the committee votes for a routine recommendation for the at-risk population, putting mpox vaccine on the immunization schedule, we very much look forward to commercializing Jynneos here in the U.S.
We have a robust manufacturing process, and we have ample supply to meet the need. We also have an unwavering commitment to public health, and we look forward to working with the agency, healthcare providers in the community to make this vaccine accessible to individuals at risk.
…
Lee Ann Kimak, Bavarian Nordic [01:43:26]
So the intended list price will be in the range of 200 to 270 dollars per dose. As you know, the list price is a price that is then negotiated down in the contracting phase, depending on the particular reimbursement mechanism. And, it comes down by 25 to 30% or more.
So this is the range for the wholesale acquisition cost.
…
Lee Ann Kimak, Bavarian Nordic [01:56:43]
We actually began taking steps towards commercializing the product as soon as we learned that the ACIP would be considering a recommendation. So if Jynneos is routinely recommended for those at risk of mpox, this would allow us to move forward with some of the steps towards commercial insurance coverage.
So we do anticipate that it will take a number of months to negotiate these contracts and of course be prepared to work through all the necessary steps with a variety of payers, setting up the distribution channels to store and administer the vaccine.
And as we’ve seen, with the commercial transition of the COVID vaccines, this process can take some time. But I wouldn’t foresee it being outside of the six to nine month range.
Agam Rao, CDC [00:01:23]
So ACIP recommends vaccination with the two dose Jynneos vaccine series for persons aged 18 years and older at risk for mpox.
And the footnotes read that this is an interim recommendation that ACIP will revisit in two to three years. That dose two has administered 28 days after dose one.
And that persons at risk include gay, bisexual, and other men who have sex with men, transgender or non-binary people, who in the past six months have had one of the following, a new diagnosis of greater than or equal to one sexually transmitted disease, more than one sex partner, sex at a commercial sex venue, or sex in association with a large public event in a geographic area where mpox transmission is occurring, or sexual partners of people with any of– with the risks described above, or persons who anticipate experiencing any of the above.
…
Grace Lee, ACIP [00:05:21]
So this vote passes a 14 yes’s, zero no’s.
October 24, 2023
CDC director at White House stakeholder briefing
- Source: webinar by White House
- Date: broadcasted October 24, 2023
Mandy Cohen, CDC [00:08:10]
This is the time when you want to get yourself vaccinated ahead of what we anticipate in the later fall and into the winter.
Right now, we’re seeing pretty low levels of COVID, flu, and RSV. We went through a COVID wave in the late summer. We had some increases in COVID in the late summer.
We had up to 30, 35,000 people in the hospital week over week at the end of August, beginning of September.
The good news is that’s coming down now, but we do expect that to go up as we get, as I said, later into the fall and into the winter.
We know when we gather inside, and we get into those colder months, we’re going to see more virus circulating.
…
Mandy Cohen, CDC [00:08:54]
As far as flu. It’s what we see every year. We’re getting into flu season now.
Flu is very low right now, but again, we expect to see that more into late November, December, January.
That is why October is the best time to get your vaccine. It allows your body to time to build up those good antibodies that protect you, so by the time you’re at Thanksgiving in just a few weeks from now, I can’t believe it here, we’re almost at the end of October, just a few weeks from now, when you’re at Thanksgiving, you’ll be fully protected with these vaccines.
…
Mandy Cohen, CDC [00:12:22]
I know we’re all wanting to move on, but I need your help to break past that fatigue, where folks want to move on with it.
It’s really important that we get folks vaccinated.
This year, we don’t have the same focus and media attention on these issues. So it’s more important than ever that we get your help.
I wish we had more, the kinds of dollars we had in the past, to spend on advertising campaigns. You’re likely not going to see that this year.
We have some of it out there and they’re focused on folks who are over 65, folks in our African American and Hispanic-Latino communities.
But you’re likely going to see less of that, which is exactly why we’re doing this today.
…
Mandy Cohen, CDC [00:14:41]
We are in close touch with folks who run long term care facilities across the country.
We were just meeting with all of those leaders here at the White House just last week. My team and myself personally am in touch with them, because we know that this is– there is a new process this year.
So in the past you likely know the federal government purchased and distributed all the COVID vaccines. This year, the private sector is in charge of that process. So it’s different this year.
So it is more challenging to get vaccines across this country and into long term care. So we’ve been working through various operational barriers and some challenges related to billing, and we continue to work closely with them to try to solve problems as we go.
But I think importantly is to get the word out.
October 20, 2023
Metadata from GISAID on JN.1 cases in the U.S.
- Source: query from GISAID’s epiCoV
- Date: searched October 20, 2023, from EPI_SET_231020hy
| Virus name | Accession ID | Collection date | Additional host information | Sampling strategy |
|---|---|---|---|---|
| hCoV-19/USA/NJ-GBW-H20-346-2416/2023 | EPI_ISL_18327656 | 9/20/23 | Traveler from Israel | |
| hCoV-19/USA/CA-CDC-LC1064285/2023 | EPI_ISL_18399956 | 9/27/23 | Baseline surveillance | |
| hCoV-19/USA/CA-Stanford-165_S10/2023 | EPI_ISL_18403077 | 9/27/23 | ||
| hCoV-19/USA/NC-CDC-LC1063242/2023 | EPI_ISL_18369238 | 9/29/23 | Baseline surveillance | |
| hCoV-19/USA/NJ-CDC-QDX85876866/2023 | EPI_ISL_18394359 | 10/2/23 | Baseline surveillance | |
| hCoV-19/USA/CA-HLX-STM-SFYSWD8NM/2023 | EPI_ISL_18407973 | 10/3/23 | ||
| hCoV-19/USA/NJ-GBW-H10-016-3356/2023 | EPI_ISL_18400009 | 10/6/23 | Airport Screening | |
| hCoV-19/USA/NJ-GBW-H20-333-0158/2023 | EPI_ISL_18400052 | 10/6/23 | Traveler from United Arab Emirates |
October 19, 2023
CDC officials at webinar on respiratory virus immunizations
- Source: webinar hosted by Centers for Disease Control and Prevention
- Date: broadcasted October 18, 2023
Question [00:47:51]
Our first chat question is an RSV question, related to nirsevimab. If a provider is unable to obtain 100 milligram dose prefilled syringes, is it acceptable to use two 50 milligram dose prefilled syringes for an infant who is five kilograms or heavier who is younger than eight months of age?
Katherine Fleming-Dutra, CDC [00:48:17]
Thank you for that question. The short answer to that is no. And the reason for that is really that we want to– the youngest infants are at highest risk for severe RSV disease. And so we want to really save those 50 milligram prefilled syringe doses for infants weighing less than five kilos.
The other concern with using two 50 milligram prefilled syringe doses for a big infant who needs a 100 milligram dose is that the prefilled syringes are the same price, 50 milligrams and 100 milligrams, and it’s not clear that insurance would reimburse a provider for using two 50 milligram prefilled syringe doses for a single infant.
So both for the reasons of making sure that we’re having enough doses to protect those youngest infants who need the 50 milligram dose, and also because of reimbursement concerns, using two 50 milligrams for a big infant is not recommended.
…
Katherine Fleming-Dutra, CDC [00:49:45]
So ideally this conversation needs to be happening with a pregnant person, with their prenatal care provider, and really it, for the vast majority of people, this will be an either or choice. So they get to choose whether or not they’d rather get the RSV vaccine during 32 through 36 weeks gestation, if it’s between September through January, most of the U.S., or have the infant receive nirsevimab after birth.
And really hopefully that prenatal provider, what we want to be happening in that setting, is that they’re having a conversation with a pregnant person talking about the risk and benefits or the relative advantages and disadvantages really of both product and making a joint decision on which is preferred for that particular individual and their family.
And so we do, I think part of that discussion can talk about, for the maternal vaccine, we know that it provides protection immediately after birth. It’s a vaccine, so there’s a broader immune response that’s being generated in mom, and those antibodies are being transferred to the baby. And so that it may be more resistant to kind of mutations in the RSV virus. It avoids injection in the infant.
But then, kind of the relative disadvantages of maternal vaccine are going to be that there are– there is this potential risk of preterm birth and hypertensive disorders of pregnancy, although FDA and ACIP feel that the benefits– judge that the benefits of vaccination outweigh the risk when used in the approved dosing interval, 32 to 36 weeks.
And then on the nirsevimab side, it provides those antibodies directly to the infant. There’s no risk of adverse pregnancy outcomes, but we do know that there’s potential limited availability, particularly this season.
So it’s really a conversation for advantages and disadvantages.
CDC director on modeling for upcoming flu season
- Source: episode of “Conversations on Health Care”
- Date: published October 19, 2023
Mandy Cohen, CDC [00:20:37]
We are able to already do a bit of modeling of what we think may happen this fall and winter.
Not surprising if all three of these viruses peak, and we have bad seasons of all. That’s not good, but we probably didn’t need a model to tell us that.
So, what we’re trying to do is with each and every passing week, we are able to sort of understand the model a bit differently and plan for the future.
So, for example, we’re sitting here in the middle of October, we already know it’s likely not to be an early flu season, which we didn’t know, because looking at the Southern hemisphere, they actually had an early flu season. We’re not seeing that here.
So that helps us again, refine the model to understand what’s going to happen over the course of the next three to four months to make sure we have the hospital beds.
So it does continue to inform our work and stay ahead to be able to plan, so we can predict and then plan.
CDC official at conference presentation on multidemic data
- Source: session at ID Week 2023 conference
- Date: broadcasted October 12, 2023
Claire Midgley, CDC [00:31:11]
CASCADIA and CoVE are just two examples of ongoing CDC funded prospective community cohorts.
CASCADIA is set out of Oregon and Washington, CoVE in Michigan, and they both started in summer of last year, roughly following individuals for kind of one to two years.
And participants have regular surveys. They have weekly swabs, regardless of symptoms, additional illness swabs, if folks do get symptomatic, and as well biennial blood draws.
The illness swabs are tested for multiple pathogens. The weekly swabs are tested for SARS-CoV-2, RSV, and flu.
And so with this kind of robust testing, they’re really able to establish and address a lot of valuable questions from COVID 19 vaccine effectiveness, to community incidence, days of school lost, other burden measures and antibody levels in the community, immunologic correlates of protection, and as well long term outcomes of infection.
…
Claire Midgley, CDC [00:42:21]
We are just about seeing RSV showing an uptick likely driven by increased circulation in the southeast United States. And if you look at the graph on the right, I’m not sure we can see the gray area, but essentially the RSV, which is the red line of this year, seems to be trending pretty closely to the gray area, which reflects the pre pandemic seasons.
And so it’s potential that RSV is moving back or close to pre pandemic circulation at this stage.
Of course, we’ll continue to monitor.
…
Claire Midgley, CDC [01:13:58]
So there’s definitely some that are already up there. RESP-NET for example, which is the hospitalization network, has a dashboard that is already kind of– Google CDC RESP-NET, there’s some– you can kind of play with ages and seasons.
And then as I mentioned, there’s also gonna be more of an influx of data this coming online this fall, in terms of being able to see different trends and some of our platforms are better positioned to really be able to look at it at the local level, like the big national platforms, like the national syndromic platform, for example, is big enough to get down to the local level.
Some of them less so. And then, in terms of actual data access, I think that’s also a part of some of the movement in trying to get more data available. Like NREVSS is one, for example, where I think where you think you can access data already.
CDC officials at Bridge Access program stakeholder briefing
- Source: recording published by Centers for Disease Control and Prevention
- Date: published October 17, 2023
Demetre Daskalakis, CDC [00:02:35]
To ensure that COVID 19 vaccines were made accessible to everyone, that’s why we had the Bridge Access program at CDC.
It was established to provide free COVID-19 vaccines to adults aged 18 years and older, who are underinsured or uninsured
Since the program began over 309,000 doses have been administered via the Bridge Access program alone and millions more through non-Bridge channels.
We want the program to be even more successful than it already has been.
…
Georgina Peacock, CDC [00:05:50]
It is important that patients with insurance understand where they are in network for vaccination and seek vaccination at those locations.
This may involve seeking vaccination within a specific system, state, or set of retail pharmacies or type of provider.
Patients do risk receiving bills for vaccination if they accidentally misrepresent their insurance coverage, when seeking vaccination through the Bridge Access program.
…
Georgina Peacock, CDC [00:07:17]
CDC is committed to ensuring everyone has access to an updated COVID 19 vaccine.
Nationally, 81% of people without insurance are estimated to be within five mile drive distance to a Bridge Access program provider.
As of October 6, there are 3,507 public health safety net providers that have placed 4,447 orders for about 205,000 doses of COVID 19 vaccine using 317 funds.
Additionally, there are currently over 23,000 contracted pharmacy locations and over 309,000 doses have been administered by contracted pharmacies to individuals without insurance.
…
Demetre Daskalakis, CDC [00:14:20]
Before you even leave to go to the pharmacy, know before you go.
So if you’re privately insured, it’s a good idea to touch base with the pharmacy or with your insurance product or plan to make sure that where you intend to get your vaccine is covered.
So the Bridge program does not cover out of network costs for individuals getting vaccinated. So if you go to a pharmacy and you’re out of network, you may incur a fee or be told that you need to go to a network pharmacy, or other site, if it’s a public health network or other type of provider.
So the best plan is that when you decide to go out to get your COVID 19 vaccine, your updated vaccine, know before you go, check with your insurance, make sure you’re going to a place that’s in network.
…
Georgina Peacock, CDC [00:18:31]
There’s always going to be some providers that are not on Vaccines.gov. Those tend to be private offices because they’re not open to the public.
But as far as Bridge Access providers, they are open and therefore need to register on Vaccines.gov so that people can find those available doses.
October 17, 2023
CDC official at presentation on wastewater disease surveillance
- Source: session at IDWeek 2023 conference
- Date: broadcasted October 12, 2023
Amy Kirby, CDC [00:26:43]
So last month was our three year birthday. So the system was established in September of 2020, and we’ve grown really rapidly. So there are 48 states, 5 cities and 2 territories that are using CDC funds to support wastewater surveillance.
We have been extremely lucky to be able to establish 4 centers of excellence that are helping us to drive forward the practice of wastewater surveillance and build, continue to build, this boat as we are sailing it. And those centers of excellence are based at a health department, and have both academic and utility partners, so that we’re really harnessing all of the disciplines that are necessary to do wastewater surveillance well.
That state led system is then supplemented by a large national testing contract that adds an additional about 400 sites, in addition to data submission from Wastewater Scan as well.
Collectively all of those sites, there’s over 1,600 sites that submit data routinely to CDC, they’re in all 50 states, 3 territories, 7 tribal communities, and collectively represent over 144 million Americans.
So we’re already collecting data every week that represents over 40% of the U.S. population.
…
Amy Kirby, CDC [00:28:11]
One of the keys to keep in mind here is that we use a distributed laboratory approach as opposed to a central lab.
So communities of course are the source of our wastewater. That goes to wastewater treatment plants. The operators there are collecting the sample and then shipping it to a laboratory, most often in their state, but sometimes in other states. And these are a collection of health department labs, academic labs, commercial testing labs, and there are a few utilities that can do this testing themselves.
That data is then sent to the health department or directly to CDC and into the DCIPHER system, which I’ll show you in just a minute, is our data repository and analysis engine. And then we analyze that data and provide it back to health departments for action and also make it available to the public.
…
Amy Kirby, CDC [00:30:25]
What our public health departments are using this for now is primarily to make decisions around resource allocation. So when they see increases in a community, they can reach out to hospitals in that community and make sure that they are prepared for the cases that will be reporting to their emergency departments in the next week or two.
They can do that either through the sort of at a glance dashboard that I was– that’s shown here, actually in the upper left. They can dive in detail. They can also compare it to other COVID measures.
…
Amy Kirby, CDC [00:31:48]
Variant tracking has been working quite well. So you can sequence SARS-CoV-2 using the same tiled amplicon approaches that you use in clinical testing.
You can do that in wastewater, but you have to use a different bioinformatics approach to analyze the data because you can’t assume that there’s only a single variant there. We know it’s probably mixed. We also know that they’re degrading in wastewater. So you have to acknowledge that there’s a bunch of puzzles that have been all split up and you’re getting little pieces of the individual puzzles, not necessarily intact genomes.
However, there are bioinformatics approaches that work well for that. So deconvolution can help you to identify variants by looking at abundance measures, as well as the sequence. And from that, we can look at what’s the dominant variant in a community. We can look at how variants have changed over time, either in that community, which is what you’re seeing there in the middle, or you can compare how it’s changed over time in that community compared to all of the other communities in that jurisdiction.
And so you can see, you know, for instance, a new variant of concern sort of moving through the communities in a way and displacing all of the prior variants.
…
Amy Kirby, CDC [00:33:58]
The most recent variant of note, BA.2.86 has really given us a new opportunity to further analyze how variant tracking can work with wastewater data.
So in addition to the deconvolution approach using Freyja that we’ve been using, you can also look specifically at a mutation based approach. So if there are unique mutations in a variant, you can look just for those, and that is a much more sensitive approach. It allows you to detect these variants even when they’re at very low concentrations.
So we’ve been doing that on a preliminary basis now, and we’re looking at how to integrate that into our pipeline for the future so that we can get the earliest detection possible of these rare variants.
…
Amy Kirby, CDC [00:40:15]
Another reason that I really think– or really am excited about pursuing these three together from NWSS is we also have anecdotal data from many of our health department partners that public health messaging that includes wastewater data gets higher engagement than messaging that just says, cases are going up in your community.
And so now that we have these tools available, the hardest thing we’re going to have to do is the behavior change to get people to actually use them. If wastewater data can help even a little bit with that by encouraging people that in your community, this is exactly what’s happening right now, that’s a win for us.
So we’re hoping to put some more formal data around that messaging story. But I think it’s worth pursuing, certainly in the meantime.
…
Amy Kirby, CDC [00:41:07]
Where are we going from here? We are continuing to build out our core surveillance. So we have pulled out the respiratory pathogens, so SARS-CoV-2, influenza A and B, and RSV, into an accelerated validation process.
So we’re going to start collecting data this fall, as soon as we can. And then, starting early next year, we’ll start validating the rest of our core panel.
So it has normalization and process controls that Allie talked about. So pepper mild mottle virus and crAssphage, which are human associated markers, bovine coronavirus, which is a process recovery control, and then we’re adding a panel of public health relevant AR threats.
So these are some of the top AR threats identified by CDC. And so we’re going to look for those.
I’m not going to talk a lot about those, because Ashley is going to talk about AR next, but we’re also going to add a small panel of foodborne infections. Those that have very low technical barriers to implementation and interpretation.
And then our emerging pathogens panel will continue to include mpox, for as long as we need to have that testing available, and we are also adding Canada Auris.
…
Amy Kirby, CDC [00:42:27]
The timeline here is quite short. So we’re looking to start piloting this with our centers of excellence at the beginning of next year. Hopefully we will have a short and problem free pilot phase, for the next six months, and then roll it out to the full system in the summer of next year.
Along with that, of course we’ll be building out DCIPHER so that we’ll have analysis and visualization and reporting tools available to our health departments, as well as building a public dashboard so that the public can use this because we do hear that people are using this to make decisions about how to keep themselves and their family safe.
CDC official at congenital syphilis presentation
- Source: session at IDWeek 2023 conference
- Date: broadcasted October 12, 2023
Lindley Barbee, CDC [00:35:57]
So these are preliminary data for 2022. They’re not finalized yet. But just like we’ve seen in the past years and through the 2021 data, we have an increase in syphilis in all persons, but primarily here, we’re looking at primary and secondary syphilis among women of reproductive age 15 to 44. And this really parallels the increase in the number of cases of congenital syphilis that you’re seeing on the right hand side in the green graph there.
Between 2013 and 2022, there was an increase from 362 cases of congenital syphilis to almost 3,500 cases for an increase of nearly 900% in the past 10 years. That also puts us at a rate of about 95 per 100 thousand live births per year.
We’re likely when we finalize this data, since this is preliminary, to reach 100 per hundred thousand live births, and this really parallels the increase in the primary and secondary syphilis rate among women of age 15 to 44, which went from 2.1 per hundred thousand persons to 17.8 in 2022.
…
Lindley Barbee, CDC [00:37:14]
In 2022, among those 3,500 cases, there were 261 deaths, including 213 stillbirths and 48 infant deaths, or about 7.5% of all reported congenital syphilis cases. And I want to remind you that this doesn’t include miscarriages because those are not reported to us.
There are over 1,200 infants who were born with signs and symptoms of congenital syphilis, and a little over 2,000 who were born with no signs or symptoms.
…
Lindley Barbee, CDC [00:37:59]
Demographics of congenital syphilis illustrates marked racial and ethnic disparities in our nation.
While the greatest number of syphilis, congenital syphilis, cases are found among Hispanic, Latino, and Black and African American populations, the highest rate of CS cases are found in American Indian and Native Alaskan natives, with the rate of almost 600 per hundred thousand live births.
The increased rate is followed by Native Hawaiians and other Pacific Islanders who have a rate of 333 per hundred thousand live births.
Our national rate, as I said on the last slide, is probably around 95 to 100 per hundred thousand live births.
I want to acknowledge that the World Health Organization has set a goal of having less than 50 per hundred thousand live births congenital syphilis cases in any given nation. We are way exceeding that right now, and some populations by nearly 10, more than tenfold.
…
Lindley Barbee, CDC [00:48:20]
Syphilis treatment is another area where we need innovation.
Recommended treatment for syphilis is penicillin, penicillin, penicillin. And while doxycycline is an acceptable alternative, it is not an acceptable alternative in pregnancy.
So there are fewer options for pregnant people and even fewer still with the current Bicillin shortage.
This is not a new phenomenon. A few years ago, we had a Bicillin shortage and we’re under another Bicillin shortage now.
We really need alternative therapies for syphilis, particularly for pregnant people.
…
Lindley Barbee, CDC [01:06:49]
CDC and FDA, you know, regularly talk to Pfizer and they are working very hard to increase production. And they have made some improvements on where they are in meeting their monthly demand.
We’re getting there, but we do need to offset a little bit of the demand for a little time period. And we are working on it, believe me, every day, all day long.
CDC official on influenza prevention, testing and treatment
- Source: session at IDWeek 2023 conference
- Date: broadcasted October 12, 2023
Tim Uyeki, CDC [00:07:04]
Right now in the U.S., there’s pretty low influenza activity. That doesn’t mean there’s zero.
We have had influenza hospitalizations in the U.S., so again, it’s not low, and we do clearly expect influenza activity to increase in the coming weeks to months.
…
Tim Uyeki, CDC [00:07:46]
So we look for some clues about what might be happening in the U.S. for this season, we typically look towards the Southern hemisphere. It’s not necessarily always a predictor, but it it’s something we wanna look at.
And interestingly enough, whereas last season in the U.S. was predominated by H3N2 viruses, what we’ve seen in the Southern hemisphere is mostly, not exclusively, but generally predominated by influenza A H1N1 pdm09 viruses.
And the reason why I mention this is it has some implications for the epidemiology.
So for H3N2 virus infections, when we see seasons predominant by that, you know, although it hits all ages, it particularly is a big impact on the elderly. A little bit less so with H1N1 pdm09 viruses.
…
Tim Uyeki, CDC [00:16:21]
Just want to say a few words about influenza home diagnostics and test to treat sites.
So there is one to date, one multiplex molecular assay that’s FDA authorized, that detects influenza A/B and SARS-CoV-2, not to advertise, but because it’s– there’s only one, it’s Lucira by Pfizer COVID 19 and flu home test.
It allows the EUA allows for collection of an anterior nasal swab for people 14 years or older, that self collection and those for two years and older, to have an adult collecting that swab.
This is for patients with signs and symptoms of respiratory tract infection.
…
Tim Uyeki, CDC [00:17:07]
There are other home influenza diagnostics in development, just to say that the availability of home influenza diagnostic tests certainly has the potential to benefit persons at increased risk of influenza complications, if it’s combined with early initiation of antiviral treatment.
So antiviral therapy has to be available. The patient has to get it, and the patient has to start taking it right away. So that could be prescribed by telemedicine or an inpatient evaluation.
And this, this actually just raises potential inequities. And so, cost, who’s going to cover the cost of the test? What’s your willingness to pay for the test? Not just the one test that’s authorized, but others to come.
I just want to raise that point.
…
Tim Uyeki, CDC [00:17:55]
And then there are a number of these test to treat sites where you can come in, particularly at pharmacies, you can get an influenza point of care test. You can get a result, and if you’re positive, you can get an antiviral drug for influenza prescribed. You can pick it up right there.
There are other test to treat projects available, not just at pharmacies, but I think this all has the potential to increase the early diagnosis and early initiation of antiviral treatment for influenza, if the patient presents early.
If the patient doesn’t present early, you have no opportunity to initiate early antiviral treatment.
…
Tim Uyeki, CDC [00:21:58]
I want to highlight a recently published study at the bottom here. This was published in June and JAMA Internal Medicine. This was a pooled meta-analysis of 15 outpatient RCTs of oseltamivir treatment in adolescents and adults. The outcome of interest was not mild disease, it was in hospitalization.
I want to make a few points because this study has gotten a lot of attention and will get some more, I think.
None of the included RCTs were actually powered for a severe outcome. They were powered for mild disease outcomes. The authors reported no association between oseltamivir and risk of hospitalization for any of the patients. And particularly for those 65 and older, or for those who were at increased risk of hospitalization.
So this my personal perspective is that this analysis was greatly underpowered. It would require an enormous sample size. The event rate for hospitalization in the untreated control group was only 0.6%.
So it’s to me inconclusive and what you really need, I don’t think anyone’s going to fund this trial, but I think what’s clearly going to solve this controversy is an enormous tens of thousands of patients would need to be recruited and enrolled to really assess this issue as question of whether oseltamivir averts the risk of severe disease requiring hospitalization.
…
Tim Uyeki, CDC [00:26:03]
One week ago, the FDA Vaccines and Related Biological Products Advisory Committee meeting voted unanimously to remove the B Yamagata antigen from the influenza vaccine, as soon as possible.
That’s probably not going to happen in the next year. It might, but just to say that globally, there’s going to be a movement from quadrivalent vaccines to trivalent vaccines by removing one of the B antigens, the B Yamagata antigen.
October 16, 2023
CDC officials at webinar with immunization programs
- Source: webinar with immunization programs by Centers for Disease Control and Prevention
- Date: broadcasted October 11, 2023
Evelyn Twentyman, CDC [00:05:18]
Specifically I wanted to share the number of doses that are going across public health infrastructure Bridge Access program sites. And that number is 3 million doses.
So this, I share this to say, this is a much smaller program than the U.S. federal government program for the distribution of COVID 19 vaccines, which ended with the onset of COVID 19 vaccine commercialization. That program was not replaced by the Bridge Access program for COVID 19 vaccines.
That program was replaced by the commercial marketplace, but it is really critical to CDC, and I think to many of you to do everything that we can to support vaccine equity, as we make the transition into the commercial marketplace. And that is why CDC worked to stand up the Bridge Access program.
Now, if all Bridge Access program vaccine doses are used, we know that we’ll achieve approximately twice the COVID 19 vaccine coverage that we did for uninsured adults in the 2022 to 2023 season. So we do hope that all of these Bridge Access program doses get used, but we did want to provide that context to help contextualize the smaller number of doses that many of the folks on this call are seeing as compared to the orders that they received in the era prior to commercialization.
This is also to say that the Bridge Access program is limited. It is temporary. It is not comprehensive in that it provides only COVID 19 vaccines. It does not provide the other vaccines, which adults are recommended to receive.
And it does not provide sufficient vaccine doses to cover the entire uninsured population, should all uninsured folks actually seek COVID 19 vaccination, the longer term and more sustainable solution to the problem of vaccine access for Americans who are uninsured, specifically American adults who are uninsured would come in the form of a Vaccines for Adults program.
I think I speak for many of us when I say that we hope that this Bridge Access program is indeed a bridge to such a more comprehensive and sustainable program. Until that point we will do everything that we can to support vaccine equity, including through the Bridge Access program.
…
Jeanne Santoli, CDC [00:10:25]
We had talked several weeks ago about Novavax’s working on a potential returns program for the public sector to sort of offset the concerns about vaccine wastage that are go along with a multi-dose vial.
And while they did come up with a returns program for the commercial customers, it ended up that they were not able to come up with a kind of efficient returns mechanism that would work for the purchase of the vaccines purchased on the CDC contracts.
So instead, what they did was to lower their price. So the vaccine that was originally, I think $72.50, was the original price that we had told you all about when we did the CNAT, actually was reduced to $58 per dose.
So it’s a significant reduction in price because that seemed the more straightforward way I think to mitigate the risk of waste. And the returns program would not have been able to sort of quickly process all of the returns.
…
Nathan Crawford, CDC [00:19:09]
Can you explain the reason that eTrueNorth cannot enroll some chains into the Bridge Access program?
I can start and then Evelyn, if you want to offer any other thoughts, this is a question we’ve received.
There are some specific requirements or expectations that provider organizations have to meet, if they’re working with eTrueNorth, and eTrueNorth can explain that to those organizations. And so that’s what we’re really recommending is that those organizations just point you point them back to eTrueNorth and they can help get them connected to make sure they can work with them.
But Evelyn, what else would you offer about that?
Evelyn Twentyman, CDC [00:19:47]
I’ll just offer that for some pharmacies, they may find themselves in some conflict with federal acquisition requirements or otherwise known as FAR requirements, if participating under one of these umbrellas.
And not being a lawyer, I’m not sure I can explain that in depth, but again, I’ll just reiterate what you so wisely said, Nathan, eTrueNorth is a great source for clarifying whether a pharmacy is able to participate in their umbrella aspect of this program or not.
…
Jeanne Santoli, CDC [00:27:21]
I am not aware of any ongoing current McKesson shipping delays. So I think if you are experiencing shipping delays that we would want you to reach out to Cameron Noblet or his team, Ashley Foss, and Kayla Artison, because we are actually not right now tracking specific shipping delays.
So I think we would want to hear from you if you have some orders that you’re concerned about. And so please reach out with specifics. That would be helpful.
…
Jeanne Santoli, CDC [00:28:34]
So we have twice now increased the amounts of Pfizer vaccine, on the 27th, I think it was sorry– on the 27th, we increased everyone by 33% and then last night we actually increased by 20%. So I think we are able to make some increases there, but what we really need people to do is place orders, get those orders out, and then we will refill them with the next cadence.
So I think there’s been some increases, but as much as you order, we will replace. So I think that’s our recommendation.
…
Evelyn Twentyman, CDC [00:30:07]
I would say that both eTrueNorth and Walgreens are able to offer popup vaccine clinics in other words, mobile clinics. And so if you wanted to get in touch with them, you could also try to work out a specific event that way, but agree with Nathan, do encourage that kind of enrollment.
…
Evelyn Twentyman, CDC [00:31:03]
While many residents of long-term care facilities do have insurance, in fact, almost all, that is not the case for long-term care facilities staff. And so to this end, actually CDC held, a convening of long-term care organizations, with ISD staff, with Dr. Mandy Cohen, with NCIRD leadership to talk about these needs and how they could be served.
Additionally, we have connected long term care facilities, with both eTrueNorth and Walgreens, to explore popups to this end.
And then on a more general note, I would say we have been relieved to see that some of the supply constraints that long term care facilities seems to have very initially, have been resolved. So the issues for residents have been resolved and resolution for staff is ongoing through CDCs Bridge Access program.
…
Evelyn Twentyman, CDC [00:35:08]
So, the way that Vaccines.gov is working has evolved with the end of the public health emergency and with the end of the way previous reporting occurred as the questioner rightly points out and clearly knows.
The issue is that we are– we have not yet built up the capacity of Vaccines.gov to onboard new providers. That is to say that providers that were not previously enrolled in Vaccines.gov are not actually currently able to enroll directly with Vaccines.gov, which does make a difference across our public health infrastructure sites.
Additionally, we are hard at work enrolling eTrueNorth en masse to allow all of these smaller and independent pharmacies to display at the same time. But that work too is ongoing.
So thanks for your patience while we sort this out.
Vaccines.gov is a collaborative effort across multiple institutions. It is not owned by CDC, but it is very important to CDC as a critical resource to others for finding these sites. So please stay tuned. We will get back to you when we know how you can enroll once again.
…
Jeanne Santoli, CDC [00:40:47]
We have allocations in place for COVID 19 vaccines. We’re actually not planning in the near term to change that because that’s the way we can try to ensure that people have equitable access to vaccines.
We’ve been able to increase several of those allocations from the initial allocations. That includes the Pfizer products and there have been two increases there. And for the Moderna pediatric, we had a significant increase there from the initial allocation.
So we’ve been able to raise the allocation levels, but we’re not in a position right now to remove allocations. So I think folks will need to be prepared for continued ordering under allocations for COVID vaccines.
…
Jeanne Santoli, CDC [00:44:43]
I think for right now, what you experienced to last night when we did the allocations across all products, is what we’re expecting will be what you will be refilled to moving forward every two weeks.
If there are increases, we’ll let you know, but we’ve had some increases to get to this point. So I think right now we– we think we might be close to where we’re gonna level off for our cadence.
…
Evelyn Twentyman, CDC [00:49:56]
We understand that there are multiple challenges with Vaccines.gov and are working through those. In fact, some of these challenges pertain to inventory and inventory, just moving really quickly within pharmacies, and pharmacies not necessarily being able to update their inventory in time to be accurately reflected on Vaccines.gov.
So this is kind of a multifactorial problem.
To be completely honest, I don’t know that we’re– that we have developed messaging to this end, but I do recommend to my patients that they simply call the pharmacy before they go. I’ve found that to be most reliable, but rest assured we are continuing to work on Vaccines.gov in the background and continuing to enable pharmacies to both have that inventory and more accurately and quickly report that inventory.
…
Jeanne Santoli, CDC [00:56:45]
I will explain the situation, Pfizer’s vaccine for children, six months to four years, does require a diluent and the diluent can’t be shipped along with the vaccine because the cold chain is different.
The vaccine coming at ultra cold and the diluent cannot be transported at ultra cold.
So the way that’s working is that the diluent ships first to ensure that you don’t get vaccine, the provider doesn’t get vaccine, without having diluent arrive either at the same time or in advance. And so because of that, they will be staggered.
They are staggered shipments and they will actually continue to be staggered shipments so that we can make sure that the diluent is there when the vaccine arrives. So I guess– I guess that might seem like a partial shipment. I was thinking of kind of only part of the vaccines arriving, but that makes sense. And so, that is actually intentional because the shipments can’t be– the products can’t be together in the same shipper.
…
Jeanne Santoli, CDC [00:58:20]
Novavax vaccines can be ordered as part of the Bridge program as well, that we have an adult contract for Novavax. So all three of those manufacturers brands would be available for the Bridge program and also for VFC because we have a pediatric contract with each of them as well.
…
Evelyn Twentyman, CDC [00:58:49]
I just wanted to add on the pharmacy component side of the Bridge Access program, only CVS will be carrying Novavax vaccine. CVS, Walgreens, and the independent pharmacies under eTrueNorth will be carrying Pfizer and Moderna.
October 13, 2023
Novavax media team on availability of their COVID-19 vaccine
- Source: email from media team at Novavax
- Date: received October 13, 2023
Novavax’s protein-based non-mRNA updated COVID-19 vaccine is now widely available across the U.S., including in pharmacies such as Costco, CVS Pharmacy, Giant, Publix, Rite Aid, Stop & Shop and Topco, physician offices and public health clinics, as well as through government entities including the Bridge Access Program and Vaccines for Children. Store vaccine locators are in the process of being updated and for any questions on availability people are encouraged to call their local pharmacy. Below is the full release from Novavax.
This follows the recent FDA authorization of Novavax’s COVID-19 vaccine in patients aged 12 years and older, developed using traditional vaccine technology. The availability of the updated Novavax vaccine provides a protein-based vaccine option offering protection against the XBB variant this fall.
Advocacy groups and pharmacists offer perspective on the availability of an additional vaccine option, noting:
“COVID continues to disproportionately impact certain communities, underscoring the persistent health disparities facing our country. The addition of another authorized COVID-19 vaccine strengthens the public vaccine supply nationwide and is another step forward in ensuring everyone has more equitable access to protecting themselves.” Sia Anagnostou, Co-Founder and Board Member, “Why We Vax”
“As a pharmacist, it’s critical that we offer patients a diverse set of COVID-19 vaccine options to help them make the right choice for themselves and their families. The option to safely mix-and-match mRNA and protein-based technologies means we can offer those in our care more choices to keep everyone safe this fall and winter.” John Beckner, R. Ph., National Community Pharmacist Association, Senior Director, Strategic Initiatives
“Timely vaccination is critical as we head into immunization season, which is why I am thrilled that families across Georgia now have an additional COVID vaccine to help protect themselves and their loved ones against COVID. We are now offering a non-mRNA, protein-based vaccine option at our pharmacy, Premier Drugstore, which uses a well-established technology used to develop other common vaccines that protect people against diseases such as influenza, hepatitis B and whooping cough. Talk with your healthcare provider or pharmacist about the option that’s right for you.” Bryan Green, Pharm.D., Owner, Premier Drugstore
If you are planning to cover this story, please let me know if I can help provide any additional information or connect you with a pharmacist, advocacy partner or company spokesperson.
CDC and AIM officials at webinar on vaccines for children
- Source: recording published by COVID-19 Vaccine Education and Equity Project
- Date: published October 11, 2023
Demetre Daskalakis, CDC [00:35:35]
We’re working really closely with all components of the supply chain, from manufacturer, distributor, etcetera, to make sure the vaccine is moving. We’ve been meeting with them actually every day, every weekday, to make sure that we are aware of how vaccine is moving, given that this is a primarily commercial vaccine.
I think from the VFC perspective, I can tell you that we’re– we have orders and shipments of over 800,000 vaccines that have already been done. So we’re actually doing gangbusters business from the perspective of VFC.
And I suspect, that early on, in the first week or two, that most of the pediatric vaccine that was in the world was actually VFC vaccine.
So, I think the other part of this is that the commercial reality of this vaccine is that providers are, I think, are starting to order more and they’re trying to sort of see how it fits into their business model as well.
And so I think that they’re seeing if this, what coverage is like, making sure that the vaccine is covered, investing what they can invest in the orders that they have coming in. So I think that really where we are able to pull the lever, which is VFC, I think that we’re doing sort of great– we’re seeing great movement of vaccine out to VFC providers and where we don’t have as much control anymore, we’re working really closely with the supply chain and with various sort of organizations that represent pediatric providers to make sure that we signal that this vaccine is covered, and in fact, that it is really a critical piece of a preventative care for their population.
…
Michelle Fiscus, AIM [00:37:24]
It’s complicated from the ground floor of all of this, because there’s so many things that have to be taken into consideration. And as Dr. Daskalakis mentioned, those pediatricians and others who vaccinate children who are doing it on the commercial side. So with, for children who are insured, it is– we have a long history of insurance companies lagging in approving payment for vaccines and other services.
And so what they’re concerned about is providing a vaccine to a child and then waiting 30, 60, 90, or forever days, for an insurance company to come back and pay them for it. And it’s quite possible that an insurance company could pay them less than they actually spent to purchase the vaccine in the first place. And that’s not a model that’s sustainable.
Pediatricians, I often refer to as the public school teachers of medicine. They’re there because they love kids. They’re not– they’re usually the lowest paid of any medical specialty. But they’re there to make sure that little kids get to grow up to be healthy adults. And that is really undervalued by insurance companies that are looking to show things on the books right now that that improve outcomes.
And sometimes when you do things for kids, those improved outcomes take 20, 30 years to show up.
So that’s going to take some time on the commercial side for pediatricians and others to feel comfortable that they’re going to get a vaccine, they’re going to give it to a child, and they’re not going to be be paying out of pocket to administer that vaccine.
…
Michelle Fiscus, AIM [00:39:08]
On the Vaccines for Children side, things are much better. And as Dr. Daskalakis is mentioned, CDC has been knocking on all the doors, and banging all the heads, trying to make sure that those VFC doses are coming in and going out.
It depends a little bit on where you live. If you’re in a very large state, that’s got tons and tons of VFC providers, there still may not be enough vaccine that are available from the manufacturers or the distributors to make sure that every single VFC office in that state has a supply of vaccines yet.
That will get better over the next few weeks as they continue to put out more vaccine. So if you can’t find it now, then just hang on and check again in a week or two, don’t give up, because it will be there.
And this is kind of an unusual situation where you’re actually going to see better access for children who are on Medicaid or uninsured than you’re going to see for children who have commercial insurance.
That’s not usually the way things work, but for a little while, we’re probably going to see that with respect to this vaccine.
October 12, 2023
HHS spokesperson on new COVID-19 vaccination figures
- Source: email from spokesperson for the Department of Health and Human Services
- Date: received October 12, 2023
“COVID-19 vaccine distribution, which has shifted to the private market, is a lot different than it was last year when the government was distributing them. The Biden-Harris Administration, through HHS, has been working directly with manufacturers and distributors to ensure that the vaccines are getting to pharmacies, hospitals, clinics, pediatricians, and other vaccination sites, including long-term care facilities. As a result, more than 7 million Americans have received their updated COVID vaccine in September, and 91% of Americans 12 years and older can access the vaccine within 5 miles of where they live. Just as we did last vaccine season, we will be working to reach Americans where they are and inform them of the updated vaccines and where they can get them.”
It is a priority for this Administration to ensure that Americans – regardless of their income or health insurance status – can access vaccines free of charge, including the uninsured through the Bridge Access Program.
Since the updated COVID-19 vaccines rollout through the commercial market, we have made the following progress to keep Americans protected this respiratory virus season:
More than 7 million Americans have received the updated COVID-19 vaccine.
Almost 14 million vaccines have shipped to pharmacies and other locations, with 91% of Americans 12 years and older living within 5-miles of a vaccination site.
And over 710,000 vaccines have been ordered through CDC’s Vaccines for Children (VFC) program, which provides vaccines to half of American children. Thirty thousand VFC sites across the country have ordered updated vaccines.
As we head into the fall and winter season, we will be encouraging Americans, particularly older adults, to get their COVID-19 vaccine in addition to their annual flu shot, the RSV vaccine for people over 60 and pregnant people during 32 and 36 weeks of pregnancy, as well as an RSV immunization for infants younger than 8 months.
October 11, 2023
FDA spokesperson on counterfeit semaglutide
- Source: email from Audra Harrison of Food and Drug Administration
- Date: received October 11, 2023
The FDA will continue to investigate each report of suspect counterfeit drugs to determine the public health risks and the appropriate regulatory response. Additionally, our Office of Criminal Investigations will continue to pursue criminal investigations targeting criminal enterprises involved in drug counterfeiting activities and other investigations with a nexus to the secure supply chain. The FDA remains vigilant in protecting the U.S. drug supply from these threats.
While we understand these drugs are in short supply and patients are having difficulty obtaining their medication, the FDA urges patients to obtain prescription drugs only from state-licensed U.S. pharmacies or physicians that are located in the United States, where the FDA and state authorities can assure the quality of drug manufacturing, packaging, distribution and labeling. FDA’s BeSafeRx campaign helps consumers understand the potential dangers of buying prescription medicines online and make informed purchasing choices. FDA urges patients to talk to their doctor if they have questions about their medications.
We also continue to raise awareness among consumers and healthcare professionals about the dangers of counterfeit drugs: Counterfeit Medicine FDA.
October 10, 2023
Ohio health department on BA.2.86 detections
- Source: email from Ken Gordon of Ohio Department of Health
- Date: received October 10, 2023
The Ohio Department of Health’s surveillance systems have found low levels of the BA.2.86 variant present in Ohio, and there remains no evidence that this variant is causing any more severe illness than other recent variants.
Our wastewater surveillance system provides early indications of changes in virus burden by geographic areas, including the presence of specific variants. At this time, we are not seeing anything of concern in any particular region as it relates to BA.2.86.
You can view our wastewater dashboard by visiting our COVID-19 data page, clicking on the “visualize” tab and then scrolling down and clicking on the “wastewater” button.
ODH continues to encourage Ohioans to stay up to date with their vaccines and to stay home if they are sick, to protect themselves and others from the virus.
October 6, 2023
FDA meeting on Southern Hemisphere influenza vaccine strain selection
- Source: livestream from Food and Drug Administration
- Date: broadcasted October 5, 2023
Jerry Weir, FDA [00:23:31]
But in addition, today, we’re going to discuss the challenges and opportunities for vaccine strain composition with the reduced public health threat from influenza B Yamagata lineage viruses.
The reason for this is kind of, sort of, twofold.
One is that, as you’ll hear probably later in the day, several times, there have been no B Yamagata lineage viruses detected for over three years now, and our VRBPAC, as in previous meetings of this committee, as well as WHO experts at the most recent strain composition meetings, have advocated for vaccine composition changes that include removal of the B Yamagata component, as well as some consideration for other composition possibilities.
…
Jerry Weir, FDA [00:29:32]
As most of you on the committee know, Dr. Wentworth has been serving as the director of the WHO collaborating center in Atlanta for several years. And he’s been making these presentations about flu vaccine composition, at least since 2019.
This will probably, I think, will be his last VRBPAC presentation in that capacity, because of his appointment recently as the director of Coronavirus and Other Respiratory Viruses Division at CDC.
So on behalf of the FDA, and for me personally, I want to thank David for all of his contributions. David has done a remarkable job of taking enormous amount of data,that the WHO sifts through for over a week and condensing it into a form that is easily explainable to all of us in about an hour. And that’s no small feat. And I think he’s done a great job over the years of doing that routinely in making all of this understandable for us.
So anyway, I would like to thank him personally, and as well as from the FDA for all he is done for us. I know the committee will thank him as well when he gets his talk, but on behalf of the FDA, I just like to say, thank you, David.
…
Jay Portnoy, VRBPAC [00:31:19]
If the committee votes no, will will there just simply not be a quadravalent vaccine, or will the B Yamagata be put in there as a placeholder anyway, given that there’s probably not enough time to come up with a different virus? What happens if we vote no?
Jerry Weir, FDA [00:31:35]
Actually I probably can’t give you a final answer today.
Obviously it’s complex, and as you will hear from the manufacturers, we’re pretty far, they will be– they will probably tell you they’re pretty far along into preparing for a Southern hemisphere vaccine, but regardless we will listen to what the committee says, and we will take that under advisement and try to formulate a plan that is both practical and useful for the Southern hemisphere.
But I do think it is important, once again, to get a sense of the committee of how they feel about this, and especially what they think about timing. So I can’t give you a final answer today.
…
David Wentworth, CDC [01:34:12]
As Dr. Weir mentioned at the outset, there have been no confirmed detections of B Yamagata circulating since after March of 2020, and this coincides with the COVID 19 pandemic.
Of the over 15,000 influenza B viruses collected between this February 1st and August 31st, this timeframe that we’re interested in, and lineage tested, no B Yamagata lineage viruses were confirmed.
…
David Wentworth, CDC [01:35:38]
It was the– our opinion as the WHO vaccine composition advisory committee that while both trivalent and quadravalent vaccines remain safe and effective, the inclusion of the B Yamgata lineage antigen in quadravalent influenza vaccines is no longer warranted, and every effort should be made to exclude this component as soon as, I put it here, practically possible.
And we can discuss that, but basically the as soon as possible was meant to be, we know there’s different cadences for different manufacturing processes. And, the fact that the committee recognizes national and regional authorities are responsible for approving the composition of formulation of vaccines used in each country and should consider the use and relative benefits of trivalent and quadravalent vaccines.
So we understand that at number one, we are supposed to make recommendations for what’s licensed. And so we make a recommendation for tri- and quadravalent vaccines, but we also wanted to make this point that we don’t think, at this point in time, it’s warranted.
It’s very difficult to determine if B Yamagata lineage viruses are really extinct. And so the, as soon as possible is meant to say, with both what needs to happen for manufacturing and quality control and for approval in the regulatory authorities, this isn’t something you do in one day.
…
David Wentworth, CDC [01:40:27]
The issue is with influenza is its speed. So as a negative strand virus, it’s poised to replicate almost as soon as uncodes inside the cell.
And I don’t want to get into the deep virology of it, but it carries all its replication machinery into the cell with it. So within six hours of infecting a cell it’s already producing progeny virion. And within overnight, an individual is now basically having millions of viruses in their respiratory tract. And they’re obviously expanding logarithmically. And then you’re transmitting influenza often before you even feel sick.
And so where the T cells become most important, particularly CD T cells, killing T-cell, is clearing the infection. So they are very important clearing the infection. But if you think about the evolution of the virus, it’s a lot– and they do impact the evolution of the virus. I don’t mean to say that they don’t, but influenza has really gone in, done its damage, and transmitted before T cells can really have a huge impact.
Remember, they’re gonna take out virally infected cells and they may be resident in your, in your respiratory airway at some level, but not at the level needed to really protect you. And so the primary driver of influenza evolution is the ability to escape those neutralizing antibodies, likely antibodies that are really IgA antibodies in the mucosal surface. Right? But, then it’s come in, done its damage, and often transmitted to many other people.
Now, the sooner T cells come in, they can actually reduce that transmission to other people in the later stages of infection. And so there is evolution at T cell epitopes that we see, but if I were to show you that on the same tree, as I show you a hemagglutinin evolution against neutralizing antibodies, the time scale is so different. Like it’s like a 10 year period, and you can start to see key epitopes in nuclear protein, as you’ve mentioned, start to be have evolution impacting them.
So I, from my perspective, I think that is saying that they do matter. They clearly matter for clearance, because folks that have immunodeficiencies with T-cells have trouble clearing influenza virus.
And that the sooner you can clear the virus, the less like you are to transmit it in the later stages of infection. And that’s how it impacts the evolution of the virus. But when we focus on the thing that has the fastest impact on the virus is the neutralizing antibodies, HA than to the NA.
So when we focus the vaccine selection more on the HA we’re really picking the tip of the spear of influenza evolution. And so by moving with that tip, you’re often updating T cell epitopes, you know, unbeknownst to you really.
…
David Wentworth, CDC [01:46:28]
This really gets outside of my area expertise on the modeling piece, what you would need to do.
What I will tell you is part of our thought process and discussions are, yeah, you could try to do this, and so, for example, I think Rinderpest would be a good, like if you really looked at how we determined Rinderpest was extinct, really good template for that, but it gets to a certain point of, it’s very hard to prove something’s extinct. Right? And what we do know is that where we’ve seen B virus, epidemics, we have not seen B Yamagata.
So it hasn’t bubbled up to cause an epidemic. We would anticipate there would be outbreaks somewhere that really across the entire global influenza surveillance system, the GISRIS, that we might pick up an outbreak, particularly in say, for example, a school where like an elementary school, where people may not have had any exposure to be B Yamagata in the past, these children, and so where it could potentially bubble up.
And so the– what we started to settle in on is what’s the real risk of being infected, not is it extinct. We don’t, we can’t, know that right now. What’s the real risk of being infected?
And the real risk from a– from our perspective is very low compared, and that’s the fact.
Now if it were to reemerge, then we can react to that, right? But standing around and really trying to do detailed studies to really prove something’s extinct takes time. And that’s the conundrum we have, I think.
We have the likely potential that it is extinct, and acting now and doing more proactive things to further improve flu vaccine, open up antigen space for other viruses, for example, in flu vaccine. And we have the potential that it is extinct.
And so why should we be growing up large quantities of virus, manufacturing, and using a vaccine that could potentially reintroduce something that’s likely extinct. And so that’s the– I think that’s what you have to weigh.
…
Andrea Shane, VRBPAC [01:49:04]
There was a percentage of B viruses that were not being able to be identified. Is there any concern that those might be Yamagata ifluenza B or not? Thank you.
David Wentworth, CDC [01:49:18]
I think we’re always going to have that issue and our concern was quite minimal on there not being.
So just to better answer Dr. Rubin’s question, one thing I do understand a little bit is trying to figure out what the level of predominance would be. And we actually have something called the rare event detection calculator on the website. It’s part of our CDC calculations for how many viruses we want to survey, how much sequencing we want to do and all that.
It’s called, calculator B, and it’s on the American Public Health Laboratories website.
But if you think about it, if we had done those extra viruses, so that would only push us from 15,000 to 20,000, and so that’s still not extensive enough to be super confident, right, that it’s gone.
So it’s reducing your ability. So for example, with that number, we know it’s likely less than 0.05% of the viruses out there in that time window with 15,000 tested. Whereas, you know, if we do another five, that’s not gonna really move that dial. We need to do an order of magnitude more to get it, to move one decimal place more basically.
And so it becomes a very costly thing and a very time consuming thing to try to do that. And you would still just be, well, it’s less than this probability.
…
David Wentworth, CDC [01:51:14]
We select these, these antigens for serologic analysis long before we have the sera. So we’re looking across the phylogeny, Dr. Kondor is very involved in this, and we’re looking to the ones that really look like they have mutations in what we know are key antigenic epitopes, and selecting those for analysis later.
And we have to grow them up, make sure they’re highly pure and sequence is good and all that. So when the human sera comes, we can do the analysis.
So what I– and they were these two A3 viruses represented by the Georgia 19. You’ve got a very good eye. They have changes in the hemagglutinin that are very significant, okay? So it’s T135K, so that removes a glycosylation site at position 133 and we know that can impact antigenicity, I140K and the S145N. So that also that 145 position is really important in antigenicity.
And so it was clear that we actually were starting to make potential candidate vaccine viruses out of that group as well, and still continue to do that, but they were somewhat deprioritized as the evolution of the virus happened, because for some reason, although it’s antigenically advanced in both ferret antisera and human antisera, it’s not winning in the world of evolution of competition of influenza viruses, the way the 3A 2A ones are.
And so we haven’t seen– it likely has some advantages of those changes, but there’s likely some fitness loss say, for example, in a non-immune host where it’s not binding receptor as well, it’s not as stable, you can think of a million things, and we don’t ever know why, but for whatever reason, it’s not increasing.
Nevertheless, it could be one that you’re discussing six months from now, when it gets an additional change, either in the hemagglutinin or a change in the neuraminidase that compensates a little bit for that fitness loss. So it likely evolved in a highly immune population, and it’s just not competing well in with all the other viruses in the population as a whole at this moment in time.
…
David Wentworth, CDC [01:55:21]
I really consider this update for the Southern hemisphere, not a major update, because really we’re staying right in that same clade of viruses. These are the most successful group. We have an antigen in Darwin six that we know is really a good antigen.
It really creates good titer, we’ve known it’s got good vaccine effectiveness for H3 viruses. And really what we’re doing is keeping the same base number of amino acid substitutions that Darwin six has, and adding that 140K, which we’ve seen in just one epitope that we’ve seen in multiple clades.
So even though it it’s a different clade of virus, say for example, the 2B with the one 140K, one would anticipate that priming the human immune system with that 140K will do better against all those viruses with the one 140K for example. And then there’s the other mutation that likely matters is at the 192.
So really it’s, it’s in some ways it’s similar to the H1. It’s not a huge antigenic difference between those two vaccines, but it’s moving a little bit closer to the influenza evolution, if you remember Darwin six was a really a really great selection in a way, because it was right during when the COVID pandemic was happening, we had very few viruses that were circulating to choose from. And that one really represented an antigenic group that looked to be expanding and created a breadth of immunity. And that’s what it turned out to be.
So it’s a little bit hard to move away from something you know is working good, but it’s what I think you have to do with influenza, because it gets you closer, it brings you closer to the virus in its evolutionary activity.
…
David Greenberg, Sanofi [02:14:25]
I’ll start by saying that we want to maintain and build public confidence and trust in influenza vaccines. We value our partnership with public health authorities, regulators, and the scientific community, and other dedicated stakeholders to provide timely access to influenza vaccines, and reduce the burden of disease worldwide.
We hear and acknowledge the ongoing concerns regarding B Yamagata.
Through this presentation, we are looking for cross-sector partnership in an organized approach that preserves public health and confidence in influenza vaccines.
While it would be helpful to have scientific regulatory and implementation frameworks to help guide the proposed removal of B Yamagata, we recognize we need to be agile and efficient in the transition. We are committed to work diligently with the FDA and other regulatory agencies worldwide.
Please also understand that our interest in documenting the rationale and framework stem from our discussions with vaccine experts in the U.S. and elsewhere, whereby they have challenged us to justify the rapid change.
…
David Greenberg, Sanofi [02:17:46]
While we’re aligned in executing the removal of B Yamagata, we recognize that transition timelines will vary based on vaccine platform, manufacturer, and various global rules, regulatory rules.
Following the WHO and MHRA meeting on July 13th, the manufacturers responded to a questionnaire to determine realistic steps for a successful global transition to TIV. And so some of those data are shown here on the slide.
There are 355 TIV licenses worldwide that need to be reactivated or submitted or resubmitted. 1,490 TIV variations needed to be updated with regulatory agencies globally. And health authorities in 174 countries will require new submissions to update manufacturing, CMC, and quality data to align with current quality standards.
…
David Greenberg, Sanofi [02:18:47]
Now, the timeframe shown here are estimates in the WHO regions based on historical standard regulatory practice. Up to 36 months in the Americas, Europe, Africa, and Western Pacific. Up to 48 months in Southeast Asia and Eastern Mediterranean.
So given our commitment to a successful transition, we look forward to receiving clear regulatory guidance and support from the FDA and agencies worldwide. We ask that the FDA provide leadership to this discussion globally and help reduce these timelines in the international markets, international regulatory agencies.
So with this global view in mind, we now want to offer specific timelines for both Southern hemisphere and Northern hemisphere transitions.
…
David Greenberg, Sanofi [02:19:54]
Given the regulatory submissions noted in the previous slide, we ask VRBPAC to make a strain recommendation for both TIV and QIV during today’s meeting, which will sustain global supply and avoid shortages.
Timelines to obtain approval for the transition from QIV to TIV will vary among national regulatory agencies throughout the world.
While TIV licenses are only inactivated in the U.S., in many countries, TIV licenses were either completely withdrawn or were never granted, thereby requiring manufacturers to submit new marketing applications that will need to be reviewed and approved.
There are Southern hemisphere countries that rely solely on CBER as the reference authority for to release vaccine. That is, we cannot ship vaccine to some Southern hemispheric countries without VRBPAC strain recommendation and FDA release.
For Southern hemisphere countries where QIV is licensed, but not TIV, we can only ship QIV. If VRBPAC does not recommend four strains, then manufacturers may not be able to supply those countries.
…
David Greenberg, Sanofi [02:21:34]
In March, while we are committed to a successful transition, it’s important to recognize that it will take some time. There is significant amount of regulatory work that was, that will be required, to transition to TIV.
Since we’re unsure of the regulatory timelines, that is manufacturers have not yet met with CBER to discuss what is needed for their specific regulatory filings, there is a substantial risk to U.S. supply if we’re faced with distributing TIV alone, just 9 to 12 months from now, that is the summer and fall of 2024.
There are important manufacturing changes that have occurred since TIV was last distributed in the U.S. and these changes require dialogue between individual manufacturers and CBER.
As of today, there’s no clear regulatory framework for reactivation for each one of the manufacturers. Those discussions are yet to come.
Based on our analysis and timeframe that I’ll show you in the next slide, in a few minutes, we propose that CBER maintained QIV licenses through this VRBPAC meeting, and when you meet again in March, we will pursue activating TIV licenses in parallel, but allowing both vaccines to coexist through these two VRBPAC sessions would be prudent and avoid supply disruptions.
…
David Greenberg, Sanofi [02:22:56]
Since the time that products were discontinued in the U.S. and internationally, many changes have occurred in the influenza vaccine manufacturing, infrastructure, quality testing, container closure, packaging, or presentation of influenza vaccines. And these changes have not been implemented with TIV.
For some products and presentations, a TIV formulation was never licensed in the U.S. Manufacturing facilities across multiple companies and external partners were built and brought online for QIV, but TIV has never been manufactured in those facilities.
End to end manufacturing, including quality and validation in many sites are QIV specific, and will need to be reevaluated and dossier submitted to regulators for TIV.
Traditionally it can take up to 18 months to generate new validation data and submit them to the agency to support licensure. So clearly we look forward to working with CBER to find a more rapid pathway for approval of TIV licenses, wherever necessary.
We should also note that when VRBPAC meets again, just five months from now to discuss the Northern hemisphere, 24-25 strain selection, most QIV orders will have already been placed by customers and manufacturing will be well underway. Therefore we ask VRBPAC to select and recommend for strains today and in March to give the industry time to obtain regulatory approvals for TIV.
…
David Greenberg, Sanofi [02:24:40] As a reminder, industry manufactures and distributes between 150 and 200 million doses of influenza vaccine for private and public sector use in the U.S. each season.
As shown, the reality is that the process for the next Northern hemisphere season is already underway. If industry manufactures only three strains and then fills and packs TIV only, but full FDA approval for the new TIV formulation is not accomplished for each manufacturer by July 2024, then U.S. supply may be compromised.
We note that customers will submit their pre-orders for QIV in the next few months. So the transition involves more than just manufacturing and regulatory approval. There’s also substantial work to manage customers, which is a complex network of healthcare providers and healthcare systems.
I’ve shared this diagram with you today to illustrate that the entire Northern hemisphere supply, or a significant portion of the supply to be shipped next summer and fall, could be lost if manufacturers produce TIV exclusively, and some unpredictable circumstance were to prohibit obtaining final regulatory approval for TIV in time.
I would like to underscore the complexity related to multiple different manufacturers and products, platforms, presentations that are necessary to fully supply the U.S. healthcare provider and system.
The significant number of unknowns that are across this complex environment requires us to keep QIV as an option for the next Northern hemisphere campaign in order for all of us to achieve our shared goal of providing an ample supply in a timely manner, allowing patients to be protected from the risk of influenza.
…
Jerry Weir, FDA [02:30:31]
Beginning in 1978, trivalent influenza vaccines incorporated, one influenza B and two influenza A, H1 and H3 components. Influenza B, though, diverged into two antigenically distinct lineages in the early 80s. And you can see a little diagram that I swiped from a Nature Communications paper that shows the timelines for these subtypes of influenza A as well as the divergence of influenza B.
Influenza B was estimated, this was after the divergence into two sub lineages, was estimated to be responsible for about 26% of influenza infections in the years 1999 to 2012. During that time, though, the mismatch frequency between the recommended strain and the most commonly circulating influenza B strain was estimated to be about 50%.
In other words, the recommendations sort of missed about half the time. I’ve thrown a couple of references for anyone that’s interested.
And so discussions began at both the WHO, VRBPAC, other places around the world in the early 2000s to discuss the feasibility and the benefit of adding a second B strain to the seasonal vaccine.
…
Jerry Weir, FDA [02:36:13]
Approval of the quadravalent vaccine was based on manufacturing and clinical data generated by each manufacturer that demonstrated safety, of course, but also the immunogenicity of the second B strain component that was added as well as that.
The addition of that second B strain did not adversely affect the immune response to other vaccine components. So that was the data package, in summary, that we used for the approval.
All currently distributed in seasonal influenza vaccines in the U.S. are quadrivalent. Basically one H1, one H3, a B Vic and a B Yam component.
…
Jerry Weir, FDA [02:36:13]
Approval of the quadravalent vaccine was based on manufacturing and clinical data generated by each manufacturer that demonstrated safety, of course, but also the immunogenicity of the second B strain component that was added as well as that.
The addition of that second B strain did not adversely affect the immune response to other vaccine components. So that was the data package, in summary, that we used for the approval.
All currently distributed in seasonal influenza vaccines in the U.S. are quadrivalent. Basically one H1, one H3, a B Vic and a B Yam component.
…
Jerry Weir, FDA [02:40:56]
In the U.S., all manufacturers of quadrivalent vaccines were originally licensed to produce trivalent vaccines, and these vaccines are still licensed, even though they’re currently discontinued.
We do have processes in the U.S., procedures for removal of vaccines from the discontinued product list. You’ve also heard that there are manufacturing changes. Some manufacturing changes have been implemented for quadravalent vaccines since the prior licensure of trivalent vaccines. We acknowledge that these changes that have been implemented for quadravalent vaccines are specific for each manufacturer.
So, other words, some may have introduced new filling lines, new presentations, others may have done other made other changes. I will point out though that in the U.S., most of these manufacturing changes that are relevant to a trivalent formulation have already been reviewed as part of the quad license. Not all, but most.
…
Jerry Weir, FDA [02:40:56]
In the U.S., all manufacturers of quadrivalent vaccines were originally licensed to produce trivalent vaccines, and these vaccines are still licensed, even though they’re currently discontinued.
We do have processes in the U.S., procedures for removal of vaccines from the discontinued product list. You’ve also heard that there are manufacturing changes. Some manufacturing changes have been implemented for quadravalent vaccines since the prior licensure of trivalent vaccines. We acknowledge that these changes that have been implemented for quadravalent vaccines are specific for each manufacturer.
So, other words, some may have introduced new filling lines, new presentations, others may have done other made other changes. I will point out though that in the U.S., most of these manufacturing changes that are relevant to a trivalent formulation have already been reviewed as part of the quad license. Not all, but most.
…
Jerry Weir, FDA [02:43:22]
First of all, there is a, a challenge in developing a consensus regarding an alternative composition. We’ve had– we have seen and heard that there are several alternative composition possibilities, and these have been proposed. I’m going to mention some of these on the next slide, but the process for making an alternative composition recommendation has not been defined.
There is also the challenge of generating the clinical data needed to support a particular alternative composition. This data would be required from each manufacturer and the data would need would be needed for each alternative composition that was proposed.
There are also technical challenges to a different composition, some of the ones that are obvious I’ve listed here, determining potency, determining identity, and then also determined to evaluating clinical outcomes. In other words, immunogenicity.
All of these are challenges because of the close relatedness of some, two different H3s, two different H1s, and some of these other combinations.
One of the other challenges that we have to keep in mind is determining the effect of an alternative composition recommendation on vaccine timelines.
With our current H1, H3, B Vic and B Yam vaccines, manufacturers have a defined process, a timeframe that they use, but they almost always prepare at least one of these antigens at risk before strain selection decisions.
So I think before anyone makes changes to the composition, we would all have to ask ourselves whether there is any effect on the timelines that result from that.
…
Jerry Weir, FDA [02:45:58]
There’s clearly still room for improvement. And that I still think presents us as an opportunity to ask ourselves whether this is a chance to actually make a vaccine that is somewhat better. Next slide.
Okay. So this is some of the other opportunities, as I said earlier, several different alternative compositions have been proposed. I listed one reference here that was a fairly recent publication that listed some of these. We had presentations at the WHO meeting in July. But you’ve also heard these over the years, all sorts of proposals.
Some of the ones that are most commonly proposed, I’ve listed in the second bullet.
For example, having two H3 antigens, sometimes you hear proposals for two H1 antigens, but here the idea would be to better cover circulating virus diversity.
There have been proposals for higher doses of H3 antigen. This would be to hopefully improve the effectiveness against the virus with the greatest public health impact.
And then there have been the proposals for higher doses of all vaccine antigens to improve the overall vaccine effectiveness.
And here, I just want to remind people that are experienced with high dose Fluzone, here with 60 micrograms of antigen, as opposed to just 15 micrograms in the standard dose, this results in the elderly with greatly improved antibody titers and improved effectiveness.
So there’s reasons to support any of these compositions.
…
Jerry Weir, FDA [02:48:10]
Every composition under consideration would need the supporting data from each manufacturer to update their license and our data needed for development for quadrivalent vaccines provides a general guide.
In other words, immunogenicity of the new component, lack of interference with other vaccine components, and safety.
…
David Greenberg, Sanofi [02:51:47]
For some Southern hemisphere countries, they rely on release by the U.S. FDA of lot by lot, batch by batch, the vaccine that can be shipped to those countries.
So although they may or may not have their own national regulatory agency, they rely on the U.S. FDA for release. So we need to continue to work with FDA in the U.S. to be able to release, let’s say, a quadrivalent to a country that does not accept trivalent yet.
…
Hana El Sahly, VRBPAC [02:53:07]
Seqirus and Sanofi are the two companies for which we meet in the fall for the Southern hemisphere in terms of FDA licensure. What is their share of the market in the Southern hemisphere?
David Greenberg, Sanofi [02:53:30]
I can begin to address that.
Our understanding that, for the two manufacturers that you just named, that there’s a approximately up to about 10 countries in Southern hemisphere where our companies ship and require the U.S. FDA release of those batches. And we’re in the range of 10 million, maybe up to 20 million, doses distributed to those countries.
And I’d say most likely the majority of them are in Latin America.
…
Jerry Weir, FDA [02:57:25]
I tried to stress that in my presentation is that there are lots of possibilities out here, but there does need to be a little focus on some of them, so that we don’t have 5 or 50 different possibilities and something that’s unmanageable.
I think there does need to be some prioritization of what we think the biggest opportunity is, and then start generating the data to make that happen.
That’s just my opinion. I don’t know if others have other comments about that over.
…
David Wentworth, CDC [02:58:37]
I’ll give you an opinion now, and it’s my opinion, not the CDC’s opinion, not WHO opinion.
It would be very challenging. I think if you went there, my thought would be, we would have to come, have a delay for a little bit and come back with what would be the alternative, like if you wanted to do an alternative strain, what would, what it would be?
At this moment in time, it’s a little tricky because it may be something like one of the two B viruses. So you’d pick a very antigenically distinct group, so the more antigenically distinct group is the two B from the two A three A1s that was nominated.
They’re actually not that far apart. And so whether or not that would be that much of an improvement, I think is the real question and why people, why the studies are needed to compare just increasing the antigen altogether versus picking two kind of divergent viruses.
If it were up to me to make the recommendation, I definitely wouldn’t pick something old and something new for the– if it was just one vaccine for every age group. And that’s because of the immune boosting that you would get to the old thing would actually probably distract from the priming that you would get from the new antigen.
And so those are the kinds of considerations that I think would have to be made now in a young population that’s maybe never seen influenza H3, that could be very valuable, right? So it’s kind of giving them an older person’s immune repertoire early in life. And, but this is really very theoretical.
And so what we work with is more like what exactly would be in this vaccine. If the committee really pushes to do something different, I think the the solution from my perspective would be to say come back with a recommendation and then we could look at that with that in mind.
…
David Wentworth, CDC [03:04:02]
Part of the B Yamagata demise is quite similar to how we have, when we have a pandemic with human influenza, it will often displace the previously circulating virus. So for example, H2N2 influenza displaced H1N1 influenza in 1957, we no longer saw H1N1 that had been circulating since 1918, very fit virus in humans.
But that even though it was very antigenically distinct, H2N2 coming in early and sweeping lots of people had a huge immune response that could be both kind of nonspecific, lots of innate immunity, et cetera. When the old virus is trying to replicate as well as recall memory response to maybe non neutralizing parts of the virus and a lot of T-cell responses to those viruses.
So I– my hypothesis really is that these two waves of distinct B Victoria lineage, even prior to the pandemic, was heavily impacting Yamagata and we really didn’t see any Yamagata, then the pandemic happened and we, again, we didn’t see very many flu viruses and what came out of the bottleneck of the pandemic, we didn’t see Yamagata again.
…
David Wentworth, CDC [03:05:48]
That’s a bit of a conjecture, a lot of conjecture about potential demise of B Yamagata, because it really started before the pandemic even happened.
…
David Wentworth, CDC [03:08:07]
The WHO global influenza program, within the pandemic preparedness and response kind of division, I’ve forgotten how they define it, which is headed by Wenqing Zhang, really does a lot of work in this space looking at gap analysis.
So where do we have gaps? How can we help? And so part of it is building an infrastructure to where, turn the lights on where it’s dark in a country that is maybe in an important area.
The one thing I would say that gives me a little bit of confidence within the regions, like if you go a little bit higher than countries and get into WHO regions, these are sometimes called transmission zones, they’re more regionally, geographically oriented, but we do have good coverage within a region.
…
David Wentworth, CDC [03:08:55]
Now you’ve pointed out Russia. And this is one of the– to Dr. Perlman’s questions, usually we have a lot more sequence data from Russia and actually have more viruses from Russia at the collaborating center at the CDC and the collaborating center at the Crick Institute in London, the worldwide influenza collaborating center there.
It’s really not– so there’s two national influenza centers in Russia, and they’re both working and collecting viruses and we are getting some sequence data out of those centers, but it’s the shipping difficulties that have led to that in this period, the last couple of years, it’s shipping difficulties that have led to some problems there.
…
David Greenberg, Sanofi [03:16:41]
I don’t have an answer for you today, but I know that there is interest among generally the manufacturing community to move in that direction. And yeah, there’s definitely work to be done, not only in understanding which would be the right, strains, say in a 3A formulation, but then also the potency testing, the immunogenicity testing, the HAIs, and so forth that all have to be worked out. So it’s also not a quick step.
And then we would in industry would conduct the clinical trials, just as we did a dozen years ago when we conducted studies of quadrivalent at the time that only trivalent was available.
So, don’t have an answer today, but we’re, I think as an industry sincerely interested and want to work with those who are here today, as well as, as others in the stakeholder community around vaccines.
…
David Greenberg, Sanofi [03:17:59]
I’m reaching back to when industry moved from trivalent to quadrivalent the probably phase one two trials may or may not be needed, but if they are, they’re probably going to be relatively small and quick. And then phase three studies are probably going to be probably in the range of hundreds of subjects, individuals who participate.
It’s not to look at efficacy, which as you know, is a much larger question, but at least, you know, confirming the non-inferior safety, or safety parameters and non-inferior immunogenicity data.
So you’re looking at a season or two to be able to conduct those studies, but perhaps that could be done efficiently in both the Southern hemisphere and the Northern hemisphere countries, so that you get to the answers needed pretty rapidly.
…
David Greenberg, Sanofi [03:19:53]
Although I don’t, again, don’t have all the answers, there could be an impact on capacity.
Having manufacturing facilities across all the different companies, running full speed for many months to produce four, working with four strains, when that reduces to three, you can see how that could, I would think reduce capacity overall then to ramp back up to quadrivalent is not difficult, but takes some planning.
So I would just remind you all that part of the rationale justification beyond the obvious of coverage of two B lineages at the time that the decisions were made, was that it increased capacity and that in the event of an influenza pandemic, it would generally be increased capacity to meet the challenges, the demand of a pandemic, with a reduction of to trivalent, you lose a little bit of that, I think we have to admit that.
…
Hana El Sahly, VRBPAC [03:22:38]
Two meetings ago, and the voting was unanimous to keep the Yamagata, but the absence of Yamagata circulation was brought up, was discussed, and pretty much everyone who voted said, this, we will keep this vote going just in case Yamagata rears its head, and to allow regulatory agencies and manufacturers time to adjust.
Last meeting, the voting was seven-six, with seven keeping the Yamagata, but even the individuals who did vote to keep the Yamagata, it was precisely to allow time for manufacturers and regulatory agencies to adjust because there’s no– the risk benefit is no longer there to keep the Yamagata.
But what I’m concerned about is that it seems like we are going to begin the discussion about it now with manufacturers and regulatory agencies. Why this not has not taken place so far and which would’ve allowed that proposed timeline to either have shifted dramatically into a smaller interval or not even be there?
…
Jerry Weir, FDA [03:25:06]
I sense and feel your frustration, Dr. El Sahly, but I’d like to remind you that the VRBPAC discussions are having an effect on moving this process, whether you believe it or not.
I actually think that this meeting that the WHO held this summer was a direct result of the voting in March. In fact, I’m pretty sure it had a lot to do with it.
So while I understand that you may be frustrated that it’s taking a while, I do think it’s having an effect. I do think it’s moving the conversation forward. And I do think that you’re going to see some tangible results for this at some point, and I hope the near future.
So again, I understand it, but I, again, I think all of you in the VRBPAC are having an effect, and you’re doing an important service by stating these concerns and stating what you think about this. I’ll just leave it at that.
…
Jerry Weir, FDA [03:28:19]
The flu community has a very unique track record of working together, unlike almost every other vaccine. We and I say we globally, and this includes manufacturers, includes this worldwide network of influenza centers, collaborating centers, ERLs, and lots of academic researchers meet periodically.
They do discuss, we all discuss these things and including we have these discussions at our advisory committee that we’re all part of.
I think this is what I meant about trying to stress the need for a consensus.
So my personal opinion is that we have these scheduled meetings at least twice a year, and this is sponsored usually by the MHRA and the WHO and we have them in July and January usually. I think that’s an ideal forum to try to get some sort of consensus of what, how one would prioritize an alternative composition, because I do think that you could list so many possibilities that you’ll never get every the data needed for everything.
So I do think that is one forum where we can do it. I think the input from the VRBPAC that we take to those meetings is important. That’s what I was trying to tell Dr. El Sahly a minute ago.
I think your voice does get heard. So I, I think this is a combined responsibility of all of us to try to work together, to see if we can make some improvement to the vaccine. I don’t know if that’s a great answer for you, but I really do believe it.
So I think we express those opinions here. We express them in these meetings where we all get together, and we hope that we drive something toward a prioritization of what should be investigated first and start generating this data. Anyway, that’s my personal view.
…
David Greenberg, Sanofi [03:50:25]
On the Northern hemisphere, for the next season, is that manufacturing has already started at risk.
Even now in October, we’re in October. So it’s, the process has started. There is a tremendous amount of coordination that occurs when we’re speaking collectively of manufacturing of more or less around 170 million doses of vaccine and decisions are made in manufacturing facilities at an early stage made right now, as to whether the final vial filling syringe filling is going to be a trivalent or a quadravalent formulation.
It’s not something that you can decide at the last minute.
So my point on behalf of manufacturing is that for the Northern hemisphere season, through the next six months, we can move as rapidly as we can to that, getting the trivalent approvals, making sure all of the validation data are with the agency and they’re reviewing them and approving them, we’re making decisions now and in the coming weeks to months on what ends up in the filling line to then ship out, starting in July.
It’s a process in parallel. We can work with the agency collectively as manufacturers to move towards trivalent. But when it, at the end of the day, when it comes to packaging filling and packaging, the product to ship, starting in July in the U.S., those decisions are made really in the next few months. So do we, as manufacturers, can we guarantee that we’re gonna have all the approvals that are necessary for reactivating the TIV and updating the quality data, and validation data to know for sure that all we need to do is package trivalent and then ship it in July, August, September? I don’t see how that’s possible across the manufacturers.
And so if we were to take that risk, then I’m really worried that we would have a major shortfall in vaccine distribution next summer and fall. So we’re going to move with the agency to get the TIV. I just don’t see, and again, speaking for all the manufacturers, just don’t see how we can guarantee that what we package in vials and syringes can only be TIV, and that we’re absolutely guarantee that we’ll be able to ship them because we haven’t had the one-on-one discussions with the agency to get us to that final point.
…
Arnold Monto, VRBPAC [03:53:44]
I was present at the July 13th meeting in London, and actually, uh, wrote the first draft of the summary of the meeting that is circulated, and Dr. El Sahly there was clearly an effect of the VRBPAC discussions and recommendations.
However, at the same time, I was rather surprised that while the global influenza regulatory and recommending community had heard the concern about the continued inclusion of the B Yamagata, the manufacturers really hadn’t heard it, and there was surprise and somewhat, I may say dismay at the possibility of moving very fast.
And that’s where we first heard some of the especially regulatory concerns.
…
Arnold Monto, VRBPAC [03:56:49]
What we’re talking about here is removing a component of the vaccine, which hasn’t been detected in greater than three years. Not only that, the component that we have in the vaccine is a 2013 virus. And if we do have B Yamagata reemerged from some hiding place, the likelihood of a B 2013 virus protecting against that virus of that strain is gonna be pretty low.
…
Arnold Monto, VRBPAC [03:58:06]
There’s been work on other things like the mRNA vaccine, but very little work on some of the simple issues about, for example, increasing the AH3N2 component of the vaccine. And I had hoped that for a smooth transition, something like that would have been looked at, but very little work has been done, or at least work that we can hear about because of other considerations.
…
Arnold Monto, VRBPAC [03:58:44]
I think it’s pretty clear that we can’t really do that right now.
The vaccine is ready to go, the manufacturing has gone on, and it would be quite disruptive to move ahead with removal right now in the Southern hemisphere formulation.
I feel uncomfortable at promising that B Yamagata lineage would be included in the Northern hemisphere formulation. I think that’s something we can take up at that time.
We may, I think it, it may be clear from our discussion in our votes that if possible, we would like to see it removed from the Northern hemisphere formulation, at least for the United States, because I think we’re in a better position in terms of the regulatory situation to do that in the United States, than many countries, which have a different system and in which the licenses for the old trivalent vaccine are not in the state that they are in the United States.
…
Arnold Monto, VRBPAC [04:00:19]
What I hadn’t thought about before the meeting in London was the fact that B Yamagata is in the live attenuated vaccine. We don’t use a whole lot of live attenuated vaccine in the United States, but there are at least theoretical disadvantages about giving a live attenuated B Yamagata containing vaccine.
And, beside that, in addition to the issue of giving something that you don’t need.
…
Hana El Sahly, VRBPAC [04:29:19]
Does the committee recommend inclusion of a B/Phuket/3073/2013 (B/Yamagata lineage)-like virus as the lineage virus as the second influenza B strain in the vaccine, please vote, yes, no, or abstain.
…
Sussan Paydar, FDA [04:36:25]
We have a total of 13 votes for question number three. Here are the responses of each of the 13 voting members for voting question number three. I’ll read themaloud for the public record. We have a unanimous vote, yes, 13 out of 13 have voted, yes.
…
Archana Chatterjee, VRBPAC [04:39:51]
I’d like to make two points, the first one being, the word, the phrase, as soon as possible.
I worked with a very experienced infection controlled practitioner, Ms. Sharon Plummer for many years at the Children’s Hospital in Omaha. And she would often say soon is not a time.
So I would say that, I would encourage all of the people that are involved to really try to identify timelines by which we should expect to see some movement on this front. So that’s the first point I’d like to make.
The second I’d like to make is that sometimes members of this committee at these meetings for the selecting the flu strains have felt that we are nearly rubber stamping what has been put forth by the WHO. So it was very reassuring to me to learn that our time is not wasted here, that in fact, our deliberations and discussions have meaning and importance and actually move the needle.
…
Arnold Monto, VRBPAC [04:41:16]
I do like the flexibility of as soon as possible, but there are timelines that have been discussed and I think we need to try to see whether it’s possible to do it in the United States, at least, by the time we meet next March, because it’s going to, it’s very clear that the whole world may not be moving in synchrony to trivalent vaccine.
…
Hana El Sahly, VRBPAC [04:52:19]
So hopefully when we reconvene in six months, the issue of the Yamagata would be behind us. That’s hopefully enough time to move on.
…
Jerry Weir, FDA [04:52:55]
My take from the discussion was that there was while everyone agreed with the ASAP as soon as possible phrase, I think everyone also realized that that was not practical for right now, the Southern hemisphere, but I did get the impression from several comments that the committee wasn’t quite ready to give up on 24-25 Northern hemisphere yet. And that we have a few months to keep working on this.
And I was pleased to hear the manufacturers say that they wanted to work with us all to see what they could do, even without any guarantees. So I think the burden is on us to do that work, to see what we can do.
I also heard that everyone was enthusiastic about trying to come up with ways to improve the vaccine with other formulations. And again, though, I got the message that what the committee would like to see is some results. Some the next time to try to hear something that’s being done to actually try to make this happen. So I think that’s another take home message that all of us heard.
And once again, I would like to thank David Wentworth for all he’s done for the last four, five years, and welcome Rebecca Kondor for future meetings like this.
CDC director at a Yale School of Public Health event
- Source: recording from Yale School of Public Health
- Date: published October 6, 2023
Mandy Cohen, CDC [00:17:02]
I think the critical linchpin of public health is to bring visibility, data and evidence to trying to solve how we protect health, whether it’s identifying and responding to a respiratory virus, or our first cases of domestically acquired malaria, which we had this year, first in 20 years, or it’s understanding overdoses from different types of fentanyl or xylazine, right.
Having that data, that transparency, is critical to building trust. So transparency for us was absolutely fundamental. And what what’s hard as, about transparency, is science is constantly evolving. As we know, we are learning more things. So it’s really critical to be able to say, this is what I know. This is what I don’t know. And importantly, this is what I’m working on to know, and I’ll come back to you, and you can trust that this public health is not a black box.
And I can’t tell you still, I was just with a round table of reporters yesterday who said that public health’s a black box to us, help us understand it better. We want to be your champions, but we don’t understand it. Let us in.
All right. So transparency, I think, builds trust. And in North Carolina, leading through the COVID crisis, we did a press conference every day at two o’clock for six months.
…
Mandy Cohen, CDC [00:44:47]
I’m so proud of CDC and only being there three months, I take no credit for this, but it is honestly the most equity focused organization that I had have been part of. And I’m very proud of the North Carolina work, but it was a lot of like new work.
When I come to CDC and I see it, the equity work is embedded everywhere in a great way. And they think about populations and they are thinking about migrants and their health at all times. And thinking about how do we make sure we’re understanding what they need and, and think about it.
So, for example, I was just on the phone with folks in New York who are seeing a lot of new immigrants and migrants that are there and how can we help them with, from an infectious disease place, but also from housing and things like that.
So CDC, like that’s, I give them a ton of credit for doing incredible work. The resources are what is missing there. It’s not the passion, it’s not the strategy, it’s resources in order to make sure that we can help all those communities.
…
Question [00:47:37]
I had a question on behalf of a person I know who’s on dialysis and they were wondering why it’s not required for healthcare workers to wear N95 respirators in healthcare settings, especially when decades of research showed that respirators are more effective than surgical masks at preventing airborne diseases. And, with like 60% of COVID infections, asymptomatic, and hospital acquired infections four times higher risk of mortality?
Mandy Cohen, CDC [00:48:11]
So, great question. So we have different teams who are looking at some of these issues right now, and I think updating some of the guidance, right? So I think you want to look at– we look at a couple of factors when thinking about it.
Sure, is an N95 gonna certainly filter out more things than a surgical mask? Yes. We know how those work, right. If you have something that’s fit tested, and the different material.
So, but the question is what are the things that you need to wear in an N95 for? And what are the things where surgical barrier masks make sense? And you want to look both at the way the infection is spread, but also what is the risk, right?
So we spread a lot of things that are are aerosolized, but there they’re different levels of risk.
It is good news in 2023, where we are related to COVID, 97% of us have either had it or have been vaccinated by it. So we aren’t in the same place we were in 2020 when we had an entire population that’s never been exposed.
Now I wouldn’t, I have to be on brand for CDC director, I’m glad we got to this, our immunity is going down. The virus is changing. Please all get your updated COVID vaccine. Even those of us who are– I’m almost was saying, I’m as young as you, I’m not, I’m getting my vaccine, but please everyone at every age should get it as well as your flu shot.
But my point is, is that we have to think about levels of risk for all of those different kinds of diseases, versus in terms of what is the protection level that we need. And so those are the kinds of research that the team goes through.
We actually have an independent advisory committee that is going through some of that work right now, to understand the data, and what, how do you map to the right protection? Because it’s both, how is it transmitted, but also like, what is the risk to all of the patients, and we sort of need to balance all of those things.
…
Mandy Cohen, CDC [00:58:24]
What I’m trying to do at CDC is make us the best collaborator we absolutely possibly could be.
Now I think data is the power behind that because, like that visualization, but it’s building a muscle of collaboration is different, right. How– like–
Candidly, CDC does not have, right now, someone who owns engagement, stakeholder engagement.
We want to call it something else, but doesn’t have that function, right? But if we’re gonna be the best collaborator and if this is a team sport, we absolutely have to build that muscle differently.
So those are the kinds of things I’m working on at CDC, but we have a lot to offer in and of itself, right? With the, the data, evidence, and best practices so that we can go to someone like CMS and be like, ‘you know what you should be paying for is X not Y.’ Or we can go to SAMHSA and say, you know what, we we’ve studied this best practice in suicide prevention, this is where you should put your money.
CMS spokesperson on nursing home COVID-19 vaccinations data
- Source: email from a CMS spokesperson
- Date: received October 6, 2023
CMS COVID-19 nursing home vaccination figures are based on information reported by nursing homes to the Centers for Disease Control and Prevention’s (CDC) National Healthcare Safety Network (NHSN) COVID-19 Long Term Care Facility Module.
On September 15, 2023, the CDC updated its recommendation regarding what is required for individuals to be up to date with their COVID-19 vaccine. The vaccination rates currently reported on data.cms.gov, as of September 24, 2023, are based on the previous recommendations. CMS is working with the CDC and plan to post vaccination rates based on the new recommendations to stay up to date with the COVID-19 vaccine soon.
For additional questions about nursing home vaccination information that reflects the current up to date recommendations, we suggest you contact the Centers for Disease Control and Prevention’s media relations staff at https://www.cdc.gov/media/subtopic/contact.htm.
Sanofi spokesperson on availability of Beyfortus
- Source: email from Sanofi spokesperson
- Date: received October 6, 2023
Sanofi has started shipping the first of its Beyfortus™ (nirsevimab-alip) doses in the U.S. to help protect infants ahead of the upcoming respiratory syncytial virus season.
…
As of October 1, coverage is in place for more than 90 percent of infant lives.
CDC officials at HRSA webinar on COVID-19 vaccines
- Source: webinar by Health Resources and Services Administration
- Date: broadcasted October 5, 2023
Evelyn Twentyman, CDC [00:17:54]
Prior to September of this year, COVID 19 vaccines were not yet on the commercial marketplace. They were bought and sold and allocated and distributed by the U.S. federal government program for COVID 19 vaccine distribution. And we are not in that era anymore.
The Bridge Access Program does not replace that program. The Bridge Access Program is a critical, but temporary and limited measure that exists to strengthen and protect the coverage of adults without insurance or whose insurance will not fully cover the cost of COVID-19 vaccination in this period, as we are transitioning into COVID-19 vaccine commercialization.
…
Evelyn Twentyman, CDC [00:18:47]
Now, how did figure out how many vaccines? That means, apart from purchasing as many as we could with the funding we were able to secure, what did that look like?
So we looked at the estimated adults who are uninsured in the U.S., and this was based on the number of adults who would be eligible in the fiscal year 24 season, which was also modified by the increase that we knew to expect in rates of uninsurance, due to Medicaid unwinding. So that’s where we got our estimation of 30 million adults who are uninsured.
And then we estimated optimistically 20% vaccine uptake per season in this population. And why am I crossing my fingers and making it look like 20% is an optimistic goal?
During the 2022-2023 season, the estimated uptake among uninsured adults, and this is pre-commercialization, in other words, this is in the era when vaccines were free to everyone, was just under 11%. So 20%, if we got 20% uptake, it would be just under twice the uptake that we saw in our preceding season.
And so if you multiply these together, you find that you need 6 million Bridge Access Program doses for the fiscal year, 2024 season, in order to provide an estimated 20% coverage of an estimated 30 million adults who are uninsured.
…
Evelyn Twentyman, CDC [00:20:45]
We did some geographic analyses, which I’m not gonna get into in super depth here, but we observed that we needed to make about a 50 50 split between public health infrastructure and retail pharmacies, or sorry, contracted pharmacies, to really maximize proximity to free COVID 19 vaccination through the Bridge Access Program.
…
Evelyn Twentyman, CDC [00:21:07]
And so there are approximately 3 million doses moving through the public health infrastructure side and approximately 3 million doses moving through the contracted pharmacy side.
3 million doses can sound like a big number until you consider the many jurisdictions this needs to cover, including the many health centers that this needs to cover. So this is a long way of saying two things.
One, these numbers are smaller than all of us are used to from the era of the U.S. federal program for COVID 19 vaccine distribution. Two, this is another way of saying this is not a permanent or comprehensive solution. It is not.
It is a critical temporary solution. It supports continued access. It is not enough.
…
Evelyn Twentyman, CDC [00:23:32]
Our current data through survey of our jurisdictions shows that allocation and distribution to these health centers is currently exceeding 25%.
So I’m going to show you two different total slides here, with one break in the middle of this data, this one is an average, and it’s over the week of September 25th through September 29th, finding that among jurisdictions who were able to respond and had data to report, 35.2% of the Bridge Access Program doses had been allocated to HRSA-supported health centers and 27.5% of Bridge Access Program doses had actually been delivered to health centers.
…
Evelyn Twentyman, CDC [00:34:21]
The important point on this question, how are we determining uninsured status, is that we designed this program to provide minimal barriers to patients seeking COVID 19 vaccination and yet maximal protection from any concerns of fraud or misuse.
And that is because all the patient needs to do is state that they are uninsured. So I’m going to go ahead and address a question right here. Do they need documentation? Do they need to somehow prove that they don’t have insurance?
No, they do not. And please do encourage your populations and your constituencies to go get this vaccine. They do not have to prove anything.
Which means they don’t need to be citizens. They don’t need to be residents. They don’t need to have these documents with them or at all.
The burden is on our health systems. So we have articulated very clearly in contracts with all of our participating providers, how they will be checking insurance status, how they will be completing the insurance discovery processes on the backend.
But this is not something that we want our community members to face.
…
Jeanne Santoli, CDC [00:36:29]
There is a, there’s a packing list that actually show the fund type of the vaccine.
So 317 is gonna be the Bridge funded doses and VFC will be the VFC doses. So that standard packing list that you get will indicate, if shipments are combined.
Pfizer doesn’t combine shipments, so they will all be separate. You won’t have to wonder which is which because they won’t combine multiple order lines in one shipment.
And I mentioned Pfizer specifically since their vaccines are actually directly shipped from the manufacturer versus the Moderna and Novavax, which are actually distributed by CDC’s centralized distributor McKesson.
…
Evelyn Twentyman, CDC [00:37:44]
It is designed to cover anyone without insurance. Now it’s not– it’s not designed necessarily for medical tourism, but in order to reduce those barriers, we’re not demanding documentation.
I know that border populations often have a lot of questions of particular interest. The important point is that barriers to vaccination are minimal and that we’re leaning on our systems, not on our patients, to document insurance.
…
Jeanne Santoli, CDC [00:41:43]
So, the orders go through the state health department because the state health departments have sort of information about the amount of vaccine that’s available. They have a certain amount of supply that’s available that they make available to providers.
They don’t do the distribution. They don’t have the doses physically on hand, but when orders come in, they manage those orders, they review those orders, and then based on that, they’re sent forward to the distributor or the manufacturer and that’s who actually sends the doses to the provider location to the health center location.
So the states have an important role, but they’re not actually doing the distribution. And that is just as it is in the VFC program.
Texas health department on genetic testing of malaria case
- Source: email from Lara Anton of Texas Department of State Health Services
- Date: received October 6, 2023
The genetic testing did not suggest a specific geographic origin for the parasite in the Texas case. It also showed that the Texas case was not a genetic match to the cluster of cases in Florida.
CDC officials at ACL webinar on COVID-19 vaccines
- Source: webinar by Administration for Community Living
- Date: broadcasted October 5, 2023
Nirav Shah, CDC [00:09:47]
I wanted to say that to acknowledge though that it’s been a challenge, but also to ask you, if you have signed up for a vaccine only to have your appointment canceled, or if you’ve been calling your doctor’s office waiting to know when the vaccine was going to be available, please don’t give up. There’s not a supply – there’s not a crunch on the supply side. There is indeed quite a bit of vaccine that’s being manufactured. Now it is working its way through the system.
So please don’t give up on vaccines. There is a vaccine out there with your name on it.
…
Amanda Cohn, CDC [00:31:48]
So if you’ve had a recent COVID vaccine or have had COVID, the infection itself, we recommend you wait about eight weeks or two months before you get that updated vaccine.
You can get it earlier, but in order to make sure you have the best immune response, we think about eight weeks is the right amount of time to wait.
…
Amanda Cohn, CDC [00:33:25]
So at this time, we’re recommending that everyone who has previously been vaccinated get one dose of this updated COVID vaccine.
We know that there may be a need for people who are severely immunocompromised to get additional doses. We are waiting to provide exact guidance on when those additional doses should happen. And you know, it would not surprise me if people who have– who are severely or moderately immunocompromised get additional dose in about six months, but right now everyone just needs one single dose.
…
Amanda Cohn, CDC [00:38:31]
Ensuring access in rural communities is especially important to us. And we totally understand that there are often people have to travel further to find vaccine, and we want to reduce that gap.
There are more Bridge Access Program providers being onboarded every day, including pharmacies health department clinics and federally qualified health centers. So I encourage you if, if you haven’t found one close enough yet, to reach back out or to reach to your health department and say, we need a location in near us, can you help us, can you help enroll?
It is our local and state health departments that are enrolling the 317 healthcare providers in this program. We are strongly encouraging the pharmacies who are involved to have a vaccine available in all locations.
But, really I think, we are relying on our local health departments to fill in some of those gaps through their healthcare providers.
…
Evelyn Twentyman, CDC [00:40:02]
If you’re in a community that does not have a close by HRSA supported health center, a CVS, a Walgreens or other 317 enrolled provider, and you do have an independent or smaller pharmacy, we do encourage you to direct them to eTrueNorth to explore participation in the Bridge Access Program.
…
Evelyn Twentyman, CDC [00:40:57]
We have two Bridge Access Program participating pharmacies who are specifically working towards setting up these community events. Those include Walgreens and eTrueNorth, and I’ve put an email for eTrueNorth in the chat and a website that contains a form to express your interest in a community event to Walgreens.
These partners of ours are very eager to work with community based organizations to get vaccine out there.
…
Amanda Cohn, CDC [00:41:27]
And I’ll just add that these– the nice thing about these pharmacies is that they can vaccinate people who have insurance as well as people who don’t, if they, as these, they put together these outreach vaccination clinics.
So in many ways, that is a really good way to make sure everyone who you serve can get vaccinated and not having to split it out into two different approaches.
American Immunization Registry Association on COVID-19 vaccine reporting
- Source: email from Rebecca Coyle of American Immunization Registry Association
- Date: received October 5, 2023
Thank you for reaching out with questions. To my knowledge I am not aware of any additional jurisdictions requiring reporting since May, but we haven’t specifically surveyed since May either.
As the federal supply of COVID-19 vaccines ends, the federal requirement to report to the IIS ends and shifts to the reporting policy of the jurisdiction. These policies vary widely from state to state.
October 5, 2023
Novo Nordisk statement on FDA investigation of counterfeit Ozempic
- Source: email from a Novo Nordisk spokesperson
- Date: received October 5, 2023
Patient safety is a top priority for Novo Nordisk and in close collaboration with FDA, we have taken measures to create awareness of the potential for counterfeit products. We have developed a company press statement that is posted on our U.S. corporate website and includes a guide for identifying counterfeits. We have provided communications to a number of stakeholders, including wholesalers and pharmacists to ensure they are aware of the situation and also able to identify a potential counterfeit semaglutide injectable product. We are also making semaglutide.com available as a resource hub for US consumers, healthcare providers and retailers to provide information about responsible use of semaglutide, including how to recognize a counterfeit product.
CDC spokesperson on locally acquired malaria in Arkansas
- Source: email from Thomas Skinner of the Centers for Disease Control and Prevention
- Date: received October 5, 2023
How many total, locally acquired malaria cases have been reported so far this year, following the report in Arkansas?
In 2023, the number of locally acquired malaria cases have included seven (7) cases have been reported in Florida, one (1) case in Texas, one (1) case in Maryland, and one (1) case in Arkansas.
Has CDC identified any links between the Arkansas malaria case and other cases?
There is no evidence to suggest the case of locally acquired malaria in Arkansas is related to the other locally acquired cases reported in the U.S. this year.
Has CDC been able to complete genomic analyses of any malaria cases so far into where the malaria cases may have originated from?
CDC is working to conduct genetic characterization of the parasites associated with these cases, including genomic sequencing, to help understand parasite relatedness and likely geographical origin. Results of sequencing are shared with the states. Please refer to the local or state health department for specific information on testing results.
Arkansas health department on locally acquired malaria infection
- Source: email from Danyelle McNeill of Arkansas Department of Health
- Date: received October 5, 2023
When was the case diagnosed? 7-10 days ago
What species caused the infection? Vivax
Was the patient hospitalized or deceased? Hospitalized, has been discharged
October 4, 2023
CDC officials at AAP webinar on fall respiratory virus season
- Source: webinar hosted by the American Academy of Pediatrics
- Date: broadcasted October 4, 2023
Mandy Cohen, CDC [00:14:07]
So we’ve passed the peak of where we think that wave at the end of the summer was, but looking at the last number of years, we expect this to go up again.
So for now we are decreasing, but again, as we get into the winter months and particularly see more travel around Thanksgiving and Christmas, we definitely do expect COVID to be up again.
We’re starting to see RSV in the Southeast, flu activity still low. So that’s where we are state of play.
…
Mandy Cohen, CDC [00:21:13]
On the ordering side, um, there’s a couple of differences with COVID 19 that that manufacturers have opened up.
You can still order in the traditional way. You’ve always ordered your vaccines in a commercial space, but at least two of the manufacturers have also opened up direct ordering, right from the manufacturer. I know Pfizer and Moderna. The Novavax vaccine just came out this week. So we’re trying to understand their ordering practices a bit more, but Pfizer and Moderna both have direct ordering opportunities. So if you are having any challenges with supply on the commercial side, through one of your distributors, so if you use one of your distributors and you’d like to order directly, you can.
There are also new changes in the return policies. So on, and different products have sort of different preparations.
On the Pfizer side, there are three doses in a vial. I know we heard concerns for example, about wastage or unused vaccine. Pfizer did change their policy just last week to say you can return any unused for a full refund. So that is also good news in terms of easing some of the ordering concerns.
On the Moderna side, they are single dose syringes. So it’s not– there’s not a waste to issue there, but again, make sure you’re talking to your distributor or the manufacturer about the rules of the road here, because they are a little bit different. Again, a bit of the feature of how we’re doing this with now that we’re working through the private sector.
…
Mandy Cohen, CDC [00:23:07]
VFC vaccines is the same way you’d order COVID as well as nirsevimab. So, nirsevimab is also able to order through the VFC program. We know that COVID 19 vaccines are shipped through CDCs central vaccine distributor or directly from the manufacturer, but say the process for ordering is still through your state or local immunization program.
And then you’ll get some email notifications again, this should seem, this should seem familiar hopefully to folks on the VFC side, but the commercial side is different, and we’re trying
…
Mandy Cohen, CDC [00:24:04]
So one of– a couple questions that we’ve been getting a lot, so I wanted to just go through this here.
First is, can I borrow between VFC and private immunization stock? And the short answer is yes, we don’t want to miss an opportunity to offer a vaccine to someone. So bidirectional borrowing is okay. We, you know, we want make sure if you borrow, that means you’re going to pay back, right? But it is allowed, but it should try to not be the rule, but you can do it.
…
Mandy Cohen, CDC [00:24:34]
The next question was can I order smaller minimums through VFC because it- and the short answer also there is yes.
We’ve decreased the minimum quantities, again, so that you can order and we’re in a pretty good– we’re getting into a better, I shouldn’t say good yet, better rhythm with ordering and then getting the supply delivered so that you can manage through how much you need in terms of understanding how many appointments you might have at a flu, COVID, vaccine clinic.
…
Mandy Cohen, CDC [00:28:09]
We certainly wouldn’t want a child to not get a vaccine because they don’t have the same– your practice doesn’t have the same type of vaccine that their initial series was in.
We have also had questions about if you do that, if pediatricians need to make reports, for example, into VAERS, and you do not need to.
So again, ideal, if it can be the same Moderna or Pfizer, but if you need to switch because of what you have available, top priority is to get these children vaccinated.
Question [00:28:37]
So that’s also true for the youngest age group, the four year olds, the six year olds?
Mandy Cohen, CDC [00:28:42]
That’s right. That’s right. So again, when you do the primary series, ideally to stick with the same manufacturer through that primary series, but if you get– then you’re into updates after that, not as much of a concern.
…
Charlene Wong, CDC [00:34:58]
This is covered in Medicaid and CHIP and VFC. Also we’ve been working with you all at the AAP and as well as with our colleagues at CMS, because we know that commercial coverage is also really important.
So, we’ve been working for example, to make sure that commercial payers have the information that they need and understand the importance of why now, coverage now, is very important in this respiratory season. So we really appreciate the partnership working with the AAP and with pediatricians across the country, so that we can all help the payers make those choices and make them quickly.
…
Georgina Peacock, CDC [00:36:31]
If you think a child is likely to have insurance, but is not yet verified, we encourage you to use your private stock. And if the child is not covered, then you can exchange it with a VFC dose. So that’s part of what we have been talking about about this bidirectional borrowing that has been given as guidance to our immunization programs.
If you expect that this child will be on Medicaid, then obviously using VFC stock makes sense. And they would be eligible for a VFC dose.
And we really understand that this is changing– it’s a difficult judgment to make in some cases. And so some of that is gonna be judgment, but hopefully that borrowing policy allows for some flexibility, so that children get the needed immunization.
…
Mandy Cohen, CDC [00:38:15]
We’re going to be communicating with our hospitals and already have about this. And, one of the questions that we were answering earlier was about, can they be VFC providers and only stock part of the VFC recommended vaccines? And the answer is yes, we want to encourage folks if they are a specialty provider, like a birthing center to be able to offer the nirsevimab and hep B at the time of birth.
And so I think this is also an education for folks and we’ll see that increase, but we’re going to try to continue like we’re doing today, to get that word out.
…
Georgina Peacock, CDC [00:38:59]
We have given guidance to our immunization program awardees that they should– that we’re encouraging them to do this, to enroll more birthing hospitals. And one of the ways to help with this is to allow virtual visits for that initial visit to set up being a VFC provider, with likely, with a follow up later that would be in person.
And so those are some flexibilities that we’ve put in the guidance and we meet regularly with our immunization awardees so that they can understand some of these flexibilities that have been put into place for niresfvimab, given that is really a very different type of product than we’ve dealt with before.
…
Mandy Cohen, CDC [00:44:00]
We are just– we are calling it an RSV immunization. It is not a vaccine in the same way in which your body creates antibodies. This immunization gives you your baby antibodies, right, they are long acting antibodies and they provide protection for about six months. So that’s the language we’re using, is that in a vaccine, it allows your body to build its own antibodies.
This RSV immunization gives the antibodies directly to your baby and they last about six months. So that’s generally the language I have been using and you will see in our materials, but Dr. Wong, anything else you’d add there?
Charlene Wong, CDC [00:44:42]
The other thing is we actually also tested with moms to see how they understood this product and you know, what, how we named it. And we actually did this in collaboration with AAP and CMS as well.
And immunization was the term that was really resonating with the moms and caregivers to these babies. So that’s a one thing to mention.
…
Mandy Cohen, CDC [00:46:26]
There are a lot of insurance companies that are covering it, but it is not, I would not say, it is all at this point. And so I would want to make sure that you are talking with your insurance companies about that coverage on the private side.
Again, for VFC, for Medicaid, that is covered. But make sure, and again, we’re continuing to work with insurance companies, and I think that we expect for them to move in that direction of coverage, but we want to make sure you’re having that conversation.
Additionally, we have not seen yet that they have changed the bundle payments for hospitals when you’re in the hospital around birth yet.
That is work that still needs to happen over the next number of months. And we expect that to happen over time, but we, that is not something for this season that we expect to see in terms of bundle, the nirsevimab being included in the bundle.
…
Charlene Wong, CDC [00:47:23]
We actually just heard, while we were talking today that, from the manufacturer, they’re letting us know that they estimate that it’s well over half of the commercial insurers that are covering nirsevimab for the outpatient setting. And, again, as we mentioned, we’re really working hard with AAP and CMS to continue to get them the information they need so that we can get that number up even higher.
…
Charlene Wong, CDC [00:51:07]
There are two administrative codes that were accepted. One is for administration of nirsevimab with counseling, by physician or other qualified health professional. And the second is that same, and that doesn’t include the counseling.
And this was really in recognition that the work that we all do at our clinics for the time to administer and the counseling is different for nirsevimab. So we’re again grateful because we worked very closely with the AAP in making sure that the codes and such really met all of the various needs.
We actually were just on a call with CMS right before this, and we’re all very excited that these codes are now available. They’re taking some of their next steps and making sure the payers will need to take some steps as, Dr. Cohen was saying both, on the particularly the commercial side. And so they’re taking some next steps right away because we did just get this very hot off the press news from the AMA just the other day. And so CMS is taking that next step, and are looking forward to having these codes available for use.
They did expedite these codes so that sometimes when these new codes become available, there’s even a longer lag before we, as providers are able to use them with the payers. They expedited this so that they become available as soon as the payers take the steps they need to take.
CDC officials at briefing on fall respiratory virus season
- Source: briefing hosted by the Centers for Disease Control and Prevention
- Date: broadcasted October 4, 2023
Nirav Shah, CDC [00:35:14]
All three channels are open and available right now, and more and more sites are being added every single day. Indeed, it’s one thing to talk about access, but we have to make sure that the pharmacy locations in particular are up and running as well as geographically spread around the country.
As of last week, there were close to 19,000 pharmacy locations, comprising both Walgreens and CVS locations across the country. And as I mentioned, more and more sites are coming online every day, particularly independent pharmacies who have partnered with our colleagues at eTrueNorth to come online to make sure that vaccine is offered in places, for example, where there isn’t as much coverage from Walgreens and CVS.
So please, please note that if there’s not a a location in your part of the part of the country right now, there may be in the near future as more and more independent pharmacies come online. That’s on the pharmacy channel.
On the provider channel more and more channels are again coming online. And they’re also increasing the administration of the vaccine that they’re offering.
As of just last week, over 6,600 pharmacy locations have administered at least one dose. And over 2,000 of the public health providers have placed at least a single order for COVID vaccines.
Again, both of those sets of numbers are increasing week on week as more providers come online and they start ordering and administering vaccine.
…
Kevin Griffis, CDC [00:42:43]
We’ve developed a suite of core messages to educate consumers around the fall and winter virus season, the importance of vaccination and other key preventative behaviors.
We know that vaccination is the best way to stay protected against flu, COVID 19 and RSV, but there are other things we can do to help prevent the spread of viruses that includes covering cough and sneezes, frequent hand washing and staying home when sick, if you’re able. We can go to the next slide.
Leaning into what we learned during our research, we developed an umbrella creative concept for our communications effort, our creative approach, which you’ll see in a moment leverages emotional ties to loved ones as the key motivator to prevent respiratory diseases, amid fall and winter gathering season, while increasing awareness and perception of severity using a holistic prevention approach to counterbalance pandemic fatigue, and concerns around fear mongering for increased receptivity educates.
It educates audiences about virus prevention behaviors, including masks and ventilation, and using icons as a tool while elevating vaccination as a key prevention strategy.
With that, I will share a preview of our efforts, which are slated to launch in mid-October. What you’ll see today is only a handful of assets that will be part of a larger effort, which with more iterations currently in development, as we continue to build out more assets that will go in the market through the end of the year.
…
Demetre Daskalakis, CDC [00:49:41]
So first I want to just put a little bit of context, which is that the largest vaccine program in the history of the U.S. transitioned on September 12th from U.S. government distribution to commercial, that’s a major milestone.
A year of planning was critical in getting ready for this. And what we learned is that public health distribution of vaccine is a lot different than commercial distribution of the vaccine. And so what we saw was a gradual increase and are seeing a day by day and week by week increase in availability of vaccine in all of the many venues that you’ve heard about, whether they’re pharmacies, medical providers, or other other folks who are vaccinators.
So, we’ve been working really closely with all other parts of government, with other agencies, but also with the manufacturers of the vaccine, as well as the distributors, to really make sure that in this very historic shift in how this vaccine is distributed, that we’re really making sure that we’re getting the vaccine to folks who need it the most, specifically, thinking about folks who are at increased risk of complications from COVID 19, specifically older individuals, as well as some younger.
So really, a lot of focus on, as we make this transition, really make sure that we’re focusing on the folks that we serve and making sure the backseat is getting where it needs to get. I’ll add on top of that, the Bridge program that you heard about in a lot of detail and Vaccines for Children have also been really critical in making sure that the vaccines that we do control in the U.S. government are getting to folks who need them.
…
Nirav Shah, CDC [00:51:19]
We’re aware that folks have perhaps gone to a provider or a pharmacy hoping to have a vaccine for themselves or for their child, and there wasn’t a vaccine there, and we’re aware of those concerns and certainly those reports.
My message for folks who are in that, who have had that experience, is twofold.
The first is please don’t give up on the vaccine. Please know that vaccine is available, and as Demetre just mentioned, more and more vaccine is getting out there every single day, every single week. And so please check back in with your provider with your pharmacist, because it’s likely if they didn’t have vaccine two weeks ago, they will have it or have it now.
The second message is please know that the CDC is working on this. When we started receiving these reports, we, as Demetre said, made sure we were working with all of the different folks who were involved in the supply chain of vaccines to see what we could do to help lift barriers, or get folks together in the same room to try to solve the problem.
The situation is getting better every single day. We know that that didn’t– we know that that meant that some folks maybe didn’t have a shot waiting for them the day that they went.
But please know that there is a vaccine out there with your name on it.
…
Kevin Griffis, CDC [00:52:41]
I know you’re planning to update the forecast and I just wanted to get a sense of if you’re seeing anything new and if there’s anything notable that people should be aware of.
Dylan George, CDC [00:52:50]
There’s a handful of uncertainties associated with trying to make these assessments going forward.
And as I mentioned, the three big uncertainties that we’re laser focused on is: is there a variant that would change our assessment of the epidemiological situation? And we’re not seeing anything like that out there right now.
The second one is, are we seeing a coincidence of the peaks of RSV, influenza and COVID coming together at some point. We’re not seeing that either.
And then the last one, are we seeing a particularly bad or severe flu season where you’re seeing a lot of people requiring hospitalization? Fortunately right now there’s low flu activity. We’re not seeing that.
But we need to stay vigilant and watch all three areas. As I had mentioned before, we’re learning more about each of these pathogens on a daily basis. As we learn more, we will update our assessments and we will update you at that time as well.
…
Nirav Shah, CDC [01:02:47]
We talk a lot about having tools this season and masking is and remains one of those tools. And I think what’s most important to think about with respect to masking is making sure that we are creating space for anyone who wants to mask to be able to do so.
Now there are a few situations in where, in which masking is important and situations that CDC recommend folks mask.
One of them, for example, is after you’ve been exposed. And all of this is on our website, but after you’ve been exposed to someone who definitely had COVID, wearing a mask after that is a great way to limit the likelihood that you might accidentally expose someone in turn, if you were to happen to contract COVID from that exposure.
Another situation where masking is recommended is on the CDC’s website, if you happen to be in a county where the rates of COVID 19 are very high, as we talk about on the website, wearing a mask is an additional layer of protection that you can take to keep yourself safe, to keep others around you safe.
…
Demetre Daskalakis, CDC [01:04:03]
So we have several strategies for really learning what’s happening in the U.S. with, not only the COVID vaccine, but other vaccines like influenza and RSV.
So through various mechanisms, through our national immunization survey, through our immunization systems that jurisdictions have, we’re able to get a view of what’s happening with vaccine.
We also, from the perspective of distribution, also know what’s happening with our Bridge program, as well as VFC, and have also been in very close contact with commercial distributors, as well as manufacturers to get a sense of how commercial distribution is also moving.
So the answer is lots of different strategies to identify how best to understand what’s happening with vaccine on the ground, in the U.S. and more to come.
As you heard from Dr. Cohen, the transparency is a really significant piece of our strategy. And, as these data become available, we’ll be sharing the best data that we have around the progress of our vaccine campaign in the U.S.
…
Demetre Daskalakis, CDC [01:07:14]
So I think as you heard from Dr. Shah already, nirsevimab is on the VFC formulary. And so it is available for VFC providers to order.
We are also in close contact with the commercial manufacturer, as well as other distributors. And the commercial component of nirsevimab is also going to be launching as we speak, now and over the next few days. So, since the majority of this immunization is gonna be distributed through the commercial market, we will continue to support the effort as well as making sure that that there’s clear guidance as to how this can be used in our youngest folks to prevent RSV.
…
Dylan George, CDC [01:08:19]
Before the end of the public health emergency ended, we were using case counts. We were using hospitalizations. And we were using death counts to anticipate what was the trajectory on both of those going forward.
Now that the public health emergency has ended, we’re focusing on hospitalization data. Those are the best data that we’re using right now to actually help us anticipate what’s going forward right now.
And we’re looking at a range of other data sources like wastewater or mobility data, or things along those lines, to see if they can be useful going forward.
But right now, the mainstay is the hospitalization data.
…
Nirav Shah, CDC [01:11:09]
With the end of the public health emergency and the commercialization of vaccines, of COVID vaccines, we don’t have the same degree of insight as to who is stocking vaccines and who’s offering them particularly at the county level.
However, the one area where we do have insight down to the provider level is through the Bridge Access Program. One of the requirements to be part of the Bridge Access Program for providers is that they list their availability of vaccine on the Vaccines dot gov website.
And so if you’re in a particular county and you’re looking for vaccine down to the zip code level, I’d urge you to go to Vaccines dot gov. That’s a starting point, particularly for those who are looking for a free vaccine from the Bridge Access Program.
If you’re looking for something a bit broader than that, you may have to check different providers websites, but if the focus is on finding a Bridge Access Program provider down to the zip code level, Vaccines dot gov is a great place to start.
…
Kevin Griffis, CDC [01:13:51]
Will there be recommendation of changes for isolation for those who are COVID 19 positive?
Demetre Daskalakis, CDC [01:13:57]
So, at this time we don’t foresee that there’s going to be a change in the isolation recommendations.
…
CDC director’s remarks at briefing on fall respiratory virus season
- Source: briefing hosted by the Centers for Disease Control and Prevention
- Date: broadcasted October 4, 2023
Mandy Cohen, CDC [00:05:12]
We’re just a few weeks into having the updated COVID vaccine, but already 4 million Americans have rolled up their sleeves in September and are better protected from COVID. That’s pretty much on par with last fall’s vaccination campaign.
We know that more than 10 million vaccines have shipped to pharmacies and other locations. And we know that Americans, now, 90% of them at least are within five miles of a vaccine site. So that’s all really good news.
…
Mandy Cohen, CDC [00:05:40]
We also have other tools to protect ourselves like tests. And we know that 42 million people have already ordered their free tests from COVID Tests dot gov, and they’re already shipping to households around the country. So that’s great news.
…
Mandy Cohen, CDC [00:07:02]
Too often, we are sacrificing understanding for precision. So we really need to continue to strive to simplify our guidance. And that is work we need to do both at the CDC, but we need your help in that to provide clear actions that people can take.
So the top line message you’re gonna hear over and over today and beyond is this.
The most effective way to protect yourself from the worst outcomes of this season’s viruses is to get your fall vaccines.
…
Mandy Cohen, CDC [00:09:34]
Equitable access to free COVID 19 vaccines going to be more challenging this season, now that the federal government doesn’t manage the whole process. Those are our realities and we have to be transparent about it, but we also have to work to address those challenges, using data and best practices and opportunities like this to build relationships with you, those who are working with our communities across the country.
October 3, 2023
From a Novavax spokesperson regarding updated COVID-19 vaccine
- Source: emails from a Novavax spokesperson
- Date: received October 3, 2023
For attribution to Novavax:
Today, the U.S. FDA authorized the first and only protein-based non-mRNA updated COVID-19 vaccine for use in people aged 12 years and older, developed by Novavax, using traditional vaccine technology. This adds a second technology option to the country’s public health arsenal to help protect individuals and their families against the XBB variant this fall. Novavax expects doses will be available in thousands of locations in the U.S., across pharmacies, physician offices and public health clinics, as well as through government entities including the Bridge Access Program and Vaccines for Children, in the coming days.
Now that EUA has been received, Novavax anticipates the U.S. Centers for Disease Control and Prevention (CDC) will issue updated recommendations for use of Novavax’s vaccine imminently.
John Jacobs, President and Chief Executive Officer, Novavax, said: “COVID-19 is once again on the rise with infections and hospitalizations increasing, so it’s important that individuals get vaccinated to protect themselves and their loved ones. Novavax’s authorization today means people will now have the choice of a protein-based non-MRNA option to help protect themselves against COVID-19, which is now the fourth leading cause of death in the U.S. In the coming days, individuals in the U.S. can go to major pharmacies, physicians’ offices, clinics and various government entities to receive an updated Novavax vaccine.”
More information on availability can be found at us.novavaxcovidvaccine.com or vaccines.gov. Press release to follow.
…
Please find below our responses and for attribution to Novavax.
When will the first Novavax vaccines be available for vaccinators? How many doses is Novavax planning to distribute this week?
Millions of doses of Novavax’s vaccine have been in U.S. warehouses since September 11. Now that we have EUA, we await CBER batch release which is anticipated in the coming days. Once batches are released, we will immediately ship and are working to have product on shelves and ready for administration immediately, pending the timing of the dose release.
Since mid-September, when the other updated COVID vaccines were greenlighted, what were the additional issues Novavax needed to address with FDA for its EUA?
Novavax worked closely with the FDA on this EUA to ensure that a protein-based option is part of the fall vaccine offering. Doses of Novavax’s COVID-19 Vaccine, Adjuvanted (2023-2024 Formula) arrived in U.S. warehouses September 11 as planned and are ready to ship to customers following CBER batch release.
From an FDA spokesperson regarding Novavax’s updated COVID-19 vaccine
- Source: email from Cherie Duvall-Jones of the Food and Drug Administration
- Date: received October 3, 2023
This action was based on the submission made to the agency by the vaccine manufacturer. Questions about the application may be directed to the company.
October 2, 2023
Official statement from Infinant Health
- Source: email from spokesperson at Leidar on behalf of Infinant Health
- Date: received October 2, 2023
I’m reaching out on behalf of Infinant Health regarding your recently published article. Please see below for an official statement from the company, which can be attributed to “An official spokesperson for Infinant Health.” If you could please confirm receipt of this email, it would be greatly appreciated.
Official Statement from Infinant Health
“At Infinant Health consumer and patient safety is our top priority. Our products are manufactured to high standards of safety and quality to deliver important nutritional benefits for full term infants.
Through ongoing discussions with the US Food and Drug Administration, we have agreed to stop shipping and voluntarily recall our probiotic Evivo with MCT Oil. That product was used by healthcare professionals in hospital settings, including neonatal care for preterm infants. FDA recently issued a warning letter objecting to the use of Evivo with MCT Oil and all other probiotics in preterm infants. We have notified medical practitioners and hospitals of our recall and the FDA warning letter.
FDA also issued a letter to healthcare practitioners advising them that probiotics can pose serious risks to preterm infants, and that the agency is investigating the death of an extremely low birth weight infant who was given Evivo with MCT Oil as part of in-hospital care. We are cooperating with FDA’s ongoing investigation. Any questions about that investigation should be directed to FDA.
Evivo powder product, available on the commercial market for consumer purchase, is not the subject of FDA’s investigation and is not being recalled. The powder product has been proven to offer significant nutritional benefits to full term infants through the gut microbiome and has been embraced by many in the pediatric care community. Evivo powder is manufactured in compliance with food safety requirements and has been certified by the Clean Label Project to help ensure its purity.
Our company of distinguished scientists, clinicians, and parents stand behind the safety and quality of our products for full term infants. We intend to work with the FDA toward approval of the use of our MCT oil product in hospital settings.”